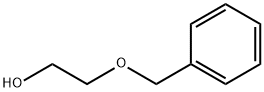
2-Benzyloxyethanol: Synthesis & Biomedical Polymer Applications
2-Benzyloxyethanol is utilized in organic synthesis for the iridium-catalyzed active methylene compounds and alcohol alkylation reactions. Due to its low volatility, it is also used as a solvent in printing inks and adhesives, as well as a fragrance fixative. Its selectivity in gas chromatography stationary liquid is similar to polyethylene glycol. 2-Benzyloxyethanol can be produced as the following procedure: Add metallic sodium to a solution of anhydrous tetrahydrofuran with ethylene glycol, maintaining a temperature of 55°C. Dropwise add benzyl chloride to the resulting mixture under reflux and let it reflux overnight. Add water, remove most of the solvent using a rotary evaporator, then add ethyl acetate to separate the organic phase. Extract the aqueous layer with ethyl acetate. Wash the combined organic layer with brine, dry with anhydrous sodium sulfate, concentrate, and purify through distillation to collect the desired product, 2-Benzyloxyethanol.

Water-Soluble and Nonionic Polyphosphoester from 2-Benzyloxyethanol
Strategies for naked DNA-based gene therapy have been widely investigated and applied in clinical trials since Wolff et al. first reported the successful expression of plasmid DNA in muscle after intramuscular injection of a foreign gene. Naked DNA, the simplest gene medicine, can be taken up by muscle cells at the DNA injection site and yields long-term expression in muscle, in some cases the expression lasting for 2 years. Poly(2-hydroxyethyl propylene phosphate) was synthesized in a method similar to poly(2-aminoethyl propylene phosphate) (PPE-EA) we described previously. A precursor polymer PPE1, obtained by ring opening polymerization of 4-methyl-2-oxo-2-hydro-1,3,2-dioxaphospholane was chlorinated according to the method described by Penczek et al., resulting in highly nucleophile-reactive P−Cl bonds. Reacting those P−Cl bonds to an excess of 2-benzyloxyethanol in chloroform using 4-(dimethylamino)-pyridine as a catalyst yielded intermediate PPE2. PPE2 is soluble in chloroform, dimethyl sulfoxide, and methanol. PPE1 (2.68 g) was suspended in 50 mL of dry chloroform.[1]
Dry Cl2 was bubbled through the mixture until the solid was completely dissolved in chloroform and a persistent yellow color appeared. The excess Cl2 and HCl gas was removed by vacuum to yield a colorless solution of poly(4-methyl-2-oxo- 2-chloro-1,3,2-dioxaphospholane). DMAP (5.34 g) was added to the above solution, followed by adding a solution of 3.68 g of 2-benzyloxyethanol in 30 mL of dry chloroform dropwise under stirring. The mixture was stirred at room-temperature overnight, and then refluxed for 2 days. The reaction mixture was washed with 1 N HCl and with distilled water three times each. The chloroform layer was dried over anhydrous magnesium sulfate and concentrated by rotary evaporation. The residue was dissolved in methanol and precipitated in ether to give PPE2 (3.95 g, yield 66%).A nonionic and water-soluble polyphosphoester, poly(2-hydroxyethyl propylene phosphate) (PPE3), was synthesized by chlorination of poly(4-methyl-2-oxo-2-hydro-1,3,2-dioxaphospholane), followed by esterification with 2-benzyloxyethanol and deprotection of the hydroxyl group by catalytic hydrogenation in the presence of Pd−C. PPE3 degraded rapidly in PBS 7.4 at 37 °C. The cytotoxicity and tissue compatibility assays suggested good biocompatibility of PPE3 in vitro and in vivo.
Tailoring thermoresponsiveness of biocompatible polyethers
Linear polyglycerol is known as a highly hydrophilic and biocompatible polymer that is currently considered for numerous medical applications. Derived from this well-known structure, the synthesis of highly biocompatible, thermoresponsive polyether copolymers via statistical anionic ring-opening copolymerization of ethyl glycidyl ether (EGE) and ethoxy ethyl glycidyl ether (EEGE) is described. Subsequent deprotection of the acetal groups of EEGE yields copolymers of linear glycerol (linG) and EGE, P(linG-co-EGE). To explore possible biomedical applications, as an example, the cell viability and immunology of an exemplary P(linG-co-EGE) copolymer sample was investigated. Since both, cell viability and immunology are comparable to the gold standard PEG, the herein presented copolymers show high potential as biocompatible and thermoresponsive alternatives to PEG for biomedical applications. The pre-dried initiator, the cesium salt of 2-benzyloxyethanol, was dissolved in DMSO-d6 and an aliquot was added to an NMR tube equipped with a Teflon stopcock. The monomers (24 mol% EEGE, 76 mol% EGE, total: 20 vol-%) were dried over CaH2 and added at −60 °C to the initiator solution. The 1H NMR spectrum of the synthesized EEGE is shown in Fig. S1 (ESI).† For copolymerizations, the initiator 2-benzyloxyethanol was deprotonated with CsOH resulting in a degree of deprotonation of 90%. The copolymers with comonomer ratios between 10 : 90 and 80 : 20 mol% (EEGE : EGE) and the corresponding homopolymers were synthesized at room temperature (25 °C) in DMSO. The herein presented copolymers are therefore suitable as a thermoresponsive alternative for mPEG in (bio)medical applications, permitting to tailor the LCST, e.g., for nanomedicine and thermoresponsive therapeutics.[2]
![Article illustration]() References
References
[1]Shi-Wen Huang. (2004). Water-Soluble and Nonionic Polyphosphoester: Synthesis, Degradation, Biocompatibility and Enhancement of Gene Expression in Mouse Muscle. Biomacromolecules, 5 2, 306–311.
[2]Müller V, Matthes R, Wagner M, Bros M, Dreier P, Frey H. Tailoring thermoresponsiveness of biocompatible polyethers: copolymers of linear glycerol and ethyl glycidyl ether. Polym Chem. 2023 Apr 14;14(21):2599-2609. doi: 10.1039/d3py00064h. PMID: 37261292; PMCID: PMC10228176.
You may like
Lastest Price from 2-Benzyloxyethanol manufacturers
US $0.00-0.00/kg2026-01-28
- CAS:
- 622-08-2
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- 500kg

US $10.00/kg2025-04-21
- CAS:
- 622-08-2
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 10 mt