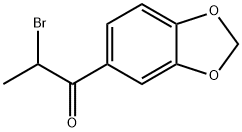
2-Bromo-3',4'-(methylenedioxy)propiophenone: Synthesis & Applications
The molecular structure of 2-Bromo-3',4'-(methylenedioxy)propiophenone incorporates both an α-bromoketone moiety and a methylenedioxybenzene ring, conferring it high reactivity. It serves as a key precursor for synthesising cathinone derivatives and MDMA analogues, while also finding application in antifungal drug development. Furthermore, it functions as a reference standard in forensic analysis and pharmaceutical research. 2-Bromo-3',4'-(methylenedioxy)propiophenone is typically synthesised from piperonyl cyclopropanone via an α-bromination reaction. Storage requires sealed containers protected from light at -20°C to prevent degradation. Note that this substance is corrosive; appropriate protective measures must be taken during handling. It is strictly for research purposes only and must not be used for any unlawful activities to ensure safe and compliant usage.

Synthesis of 2-Bromo-3',4'-(methylenedioxy)propiophenone
Psychedelics are substances inducing unique subjective effects including dream-like alterations of consciousness, affective changes, enhanced introspective abilities, visual imagery, pseudo-hallucinations, synesthesia, mystical-type experiences, ego-dissolution, and feelings of connectedness. The present invention provides for a composition of a compound represented for use in treating medical disorders, wherein Rai and R<X2 is, independently and in any combination, hydrogen, deuteron, C1-C6 saturated and unsaturated alkyl optionally deuterated or fluorinated, C3-C6 saturated and unsaturated cycloalkyl-(Ci-C6)alkyl optionally deuterated or fluorinated, and Rai and Ra2 can be combined to form a cyclic moiety such as cycloalkyl, oxacycloalkyl, thiacycloalkyl or azacycloalkyl, which can be further substituted in any combination with deuteron, fluorine, alkyl, alkenyl or alkynyl substituents. Preparation of the allylamine-type phenethylamine derivative involves synthesis of 2-Bromo-3',4'-(methylenedioxy)propiophenone. Bromine (5.1 mL; 100mmol) was added dropwise under occasional cooling to a solution of 17.82g (100 mol) 1 -(1 ,3-benzodioxol-5-yl)propan-1 -one in 160mL dichloromethane (DCM), whereby large amounts of HBr gas evolved, which were trapped in a gas washing bottle containing ice-cooled aqueous NaOH. After 1 hour the reaction mixture was poured into 400mL icewater, and the organic layer was washed with water (4x100mL), dried over Na2SO4, and concentrated in vacuo. Yield: 23.98g (93.3%)2-Bromo-3',4'-(methylenedioxy)propiophenone as a brown solid.1 H-NMR (CDCI3): 1 .91 (d, CH3 ), 5.22 (q, BrCH), 6.09 (s, OCH2O), 6.90 (d, 1 arom. H), 7.51 (d, 1 arom. H), 7.67 (dxd, 1 arom. H).
2-Bromo-3',4'-(methylenedioxy)propiophenone applied in talaumidin derivatives
Scientists established a new synthetic methodology for talaumidin derivatives in order to explore new neurotrophic compounds that can be obtained on a large scale. In addition, we discuss the structure-activity relationships between talaumidin derivatives and neurotrophic activities. To a solution of 1-(4-Benzyloxy-3-methoxyphenyl)propan-1-one (3.90 g, 14.4 mmol) in CHCl3 (100 mL) was added Br2 (741 μL, 14.4 mmol) in CHCl3 (45.0 mL) at 0 °C. After being stirred overnight, saturated NaHCO3 solution was added and extracted with DCM. The organic layers were dried over Na2SO4 and concentrated in vacuo. The residue was purified by column chromatography (hexane:EtOAc = 88:12) to afford 2-Bromo-3',4'-(methylenedioxy)propiophenone (3.93 g, 78%) as a colorless oil. 1H NMR (300 MHz, CDCl3) δ 7.59 (1H, brs), 7.58 (1H, brd, J = 8.2 Hz), 7.44–7.31 (5H, m), 6.90 (1H, d, J = 8.2 Hz), 5.25 (1H, q, J = 6.6 Hz), 5.23 (2H, s), 3.94 (3H, s), 1.87 (3H, d, J = 6.6 Hz). 13C NMR (75 MHz, CDCl3) δ 192.1, 152.9, 149.7, 136.1, 128.7, 128.2, 127.2, 123.2, 112.0, 111.5, 70.8, 56.1, 41.2, 20.4. IR (ATR) 2933, 2358, 1671, 1591, 1453, 1376, 1260 cm−1. HRMS (EI) m/z: calcd. for C17H17O3Br [M]+ 348.0362, found 348.0356.[2]
Compound 1-(benzo[d][1,3]dioxol-5-yl)-2-bromopropan-1-one was prepared in the same manner as compound 2-Bromo-3',4'-(methylenedioxy)propiophenone using ketone (9.00 g, 50.0 mmol). The product 1-(benzo[d][1,3]dioxol-5-yl)-2-bromopropan-1-one (11.3 g, 87%) was obtained as a pale yellow oil. 1H NMR (300 MHz, CDCl3) δ 7.64 (1H, dd, J = 8.1, 1.8 Hz), 7.50 (1H, d, J = 1.8 Hz), 6.88 (1H, d, J = 8.1 Hz), 6.07 (2H, s), 5.22 (1H, q, J = 6.6 Hz), 1.88 (3H, d, J = 6.6 Hz). Compound 2-Bromo-1-(4-methoxyphenyl)propan-1-one was prepared in the same manner as compound 2-Bromo-3',4'-(methylenedioxy)propiophenone using ketone α-bromoketone (2.00 g, 12.2 mmol). The product 2-Bromo-1-(4-methoxyphenyl)propan-1-one (2.45 g, 83%) was obtained as a pale yellow oil. 1H NMR (300 MHz, CDCl3) δ 8.01 (2H, d, J = 9.3 Hz), 6.95 (2H, d, J = 9.3 Hz), 5.26 (1H, q, J = 6.6 Hz), 3.88 (3H, s), 1.89 (3H, d, J = 6.6 Hz). To a solution of 6a (1.00 g, 5.58 mmol) in DMF (35.0 mL) was added KHMDS (13.4 mL, 0.5 M solution in toluene) at −45 °C. After being stirred for 1 h, a solution of 2-Bromo-3',4'-(methylenedioxy)propiophenone (1.95 g, 5.58 mmol) in DMF (35.0 mL) was added to the reaction mixture at the same temperature. After being stirred overnight, the reaction was quenched with saturated NH4Cl. The aqueous layer was extracted with ether. The combined organic layers were washed with brine, dried over anhydrous MgSO4, and concentrated in vacuo. The residue was purified by column chromatography (hexane:ether = 1:1) to afford 1-(benzo[d][1,3]dioxol-5-yl)-4-(4-benzyloxy-3-methoxyphenyl)-2,3-dimethylbutane-1,4-dione (2.15 g, 86%) as a pale yellow oil.
References
[1]WO2024263978
[2]Harada, K., Zaha, K., Bando, R., Irimaziri, R., Kubo, M., Koriyama, Y., & Fukuyama, Y. (2018). Structure-activity relationships of talamidin derivatives: Their neurite-outgrowth promotion in vitro and optic nerve regeneration in vivo. European Journal of Medicinal Chemistry, 148, 86-94.
You may like
Lastest Price from 2-Bromo-3',4'-(methylenedioxy)propiophenone manufacturers
US $0.00-0.00/kg2025-12-08
- CAS:
- 52190-28-0
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- 1000kg
![52190-28-0 1-(Benzo [d][1, 3] dioxacyclopentene-5-yl) -2-bromoprop-1-ketone](/ProductImageEN1/2025-02/Small/602de116-b584-4e5c-8a1b-4fad0b1cc3ea.jpg)
US $100.00-50.00/KG2025-03-21
- CAS:
- 52190-28-0
- Min. Order:
- 1KG
- Purity:
- 99.99%
- Supply Ability:
- 5T