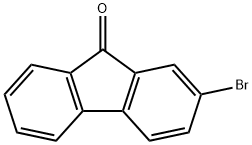
2-Bromo-9-fluorenone: Fluorenone Derivative & Photocatalyst
2-Bromo-9-fluorenone is a yellow crystalline solid at room temperature. Its chemical structure, featuring a fluorene backbone substituted with a bromine atom and a ketone functional group, imparts unique reactivity and desirable photophysical characteristics. The synthesis of 2-Bromo-9-fluorenone is most commonly achieved through the oxidation of 2-bromofluorene. Various methods have been developed to optimize yield and purity.

Diastereoselective and Stereodivergent Synthesis of C-Styryl Glycosides
A novel and efficient method for the stereoselective and stereodivergent synthesis of both unprotected and protected styryl C-glycosides through the reaction of sodium glycosyl sulfinate and hypervalent styryl iodine reagents is described. Using 2-bromo-9-fluorenone as a photocatalyst, C-styryl glycosides with an E configuration were selectively obtained. Furthermore, the introduction of a newly designed iridium-based catalyst enabled the selective formation of glycosides predominantly in the Z configuration. Mechanistic studies reveal the glycosyl radical is involved in the transformation. Using photocatalyst 2-bromo-9-fluorenone selectively yields products with the E configuration. Furthermore, a newly developed iridium-based photocatalyst enabled the selective formation of Z-configured glycosides. Mechanistic studies reveal a unique π-complex between hypervalent iodine and the fluorenone photocatalyst, which facilitates the efficient generation of the glycosyl radical via a SET process for the following alkenylation. This work not only expands the synthetic tool kit for stereoselective glycosylation but also uncovers a novel glycosyl radical generation pathway.[1]
Solubility of 2-Bromo-9-fluorenone in 10 Different Organic Solvents
In recent years, fluorenone and its derivatives have become a class of important functional compounds. 2-Bromo-9-fluorenone (2B9F), as a derivative of fluorenone, is used to synthesize intermediates of functional polymers, medicines, pesticides, and photosensitive dyes. For example, according to the report of Anand S, 2B9F can be used to synthesize anticancer drugs, sympathetic depressants, and antispasmodic agents in the pharmaceutical industry. Hsu presented that 2B9F can synthesize herbicides and insecticides. In addition, it is also used to synthesize aromatic diamine dyes and photosensitive materials in the dye industry and the classical photocopying industry, respectively. In the plastics industry, 2-bromo-9-fluorenone can be used for the synthesis of bisphenol products, stabilizers, and plasticizers. To achieve large-scale production of 2B9F, it is necessary to find a suitable solvent for recrystallization. Due to 2B9F having a lower solubility in aqueous solution, common industrial organic reagents were chosen as the research model, such as alcohols (methanol and ethanol) and ester (ethyl acetate). Additionally, in this work, because 2-bromo-9-fluorenone is a conjugated aromatic hydrocarbon with a complex chemical structure, 1,4-dioxane with strong dissolving power for most drugs was selected as the co-solvent and methanol, ethanol, and isopropanol were selected as the antisolvent; the co-solvent and the antisolvent were mixed in a certain proportion.[2]
In this work, the solubility of 2-bromo-9-fluorenone in 10 pure solvents (methanol, ethanol, n-propanol, isopropanol, n-butanol, acetonitrile, methyl acetate, ethyl acetate, butyl acetate, 1,4-dioxane) and three binary solvent mixtures has been determined. The order of solubility of the 10 pure solvents was 1,4-dioxane > butyl acetate > ethyl acetate > methyl acetate > n-butanol > acetonitrile > n-propanol > isopropanol > ethanol > methanol. In three binary solvent mixtures, the order of solubility was methanol + 1,4-dioxane <ethanol + 1,4-dioxane < isopropanol + 1,4-dioxane. The maximum value of 2B9F solubility data is 0.0526 (mole fraction) in 1,4-dioxane at 323.15 K, and the minimum value is 0.0002 (mole fraction) in methanol at 278.15 K. Therefore, 1,4-dioxane could be selected as an ideal solvent for industrial separation and purification of 2-bromo-9-fluorenone. By comparison, the modified Apelblat model was significantly better than the Buchowski–Ksiazaczak λh model in fitting the solubility data for pure solvents, and the CNIBS/R-K model was superior to the Jouyban–Acree model in fitting the solubility in binary solvent mixtures. The solubility was negatively correlated with the polarity of the solvent. It was found that the hydrogen bond had little effect on the solubility of 2-bromo-9-fluorenone by the KAT-LSER model, and the cohesion energy density was the most important factor. Therefore, the solubility data of this experiment can provide a reference for the separation and purification of 2B9F and guide the large-scale production of many drugs.
References
[1]Ma, Jialu et al. “Photoinduced Diastereoselective and Stereodivergent Synthesis of C-Styryl Glycosides.” Organic letters vol. 27,21 (2025): 5458-5463. doi:10.1021/acs.orglett.5c01437
[2]Yanjuan Peng, & Li Mi*, (2022). Determination and Analysis of Solubility of 2-Bromo-9-fluorenone in 10 Different Organic Solvents and Three Binary Solvent Mixtures at Different Temperatures (T = 278.15–323.15 K). Journal of Chemical & Engineering Data, 67 9, 2662–2674. https://doi.org/10.1021/acs.jced.2c00163
You may like
Lastest Price from 2-Bromo-9-fluorenone manufacturers
US $0.00/kg2025-12-17
- CAS:
- 3096-56-8
- Min. Order:
- 100kg
- Purity:
- 99.8~99.98%
- Supply Ability:
- 50t

US $0.00-0.00/Kg/Drum2025-04-21
- CAS:
- 3096-56-8
- Min. Order:
- 1KG
- Purity:
- 99%min
- Supply Ability:
- 1000kg