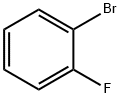
2-Bromofluorobenzene: Cross-Coupling & Heterocycle Synthesis Applications
2-Bromofluorobenzene is primarily employed as an intermediate in pharmaceuticals and pesticides. It is a chemical substance composed of multiple raw materials that is harmful if ingested and irritates the eyes, respiratory system, and skin. Current production methods for it generally rely on external containment, with no effective measures implemented for exhaust gas emissions. 2-Bromofluorobenzene serves as a thickener, tackifier, emulsifier, stabiliser, moulding agent, and suspending agent, finding extensive application across petroleum, chemical, food, papermaking, light industry, textile, dye, pharmaceutical, agricultural, and ceramic sectors. Presently, this kind of products are chiefly utilised within the food industry to diversify product ranges, enhance quality, and elevate food standards.

Suzuki-Miyaura C-C Coupling Reactions
Palladium-catalyzed reactions have been one of the main methods for C-C cross-coupling processes. In particular, Suzuki-Miyaura are one of the most widely used reactions for the preparation of biphenyl derivatives. These reactions have been principally carried out using homogeneous catalysts based on simple or sophisticated Pd complexes. However, the current needs of industry and the search for greener alternatives to these catalysts are pushing the development of new heterogeneous and recyclable systems. These heterogeneous C-C coupling catalysts are based on either supported palladium complexes or supported palladium nanoparticles. In this context, our group has decided to study the preparation of different fluorinated biphenyl derivatives by Suzuki-Miyaura coupling reactions. In general, fluorinated compounds, although generally viewed as mostly inert because of their lack of chemical reactivity, may have biological activity which could be of interest in different therapies. In view of the interesting catalytic properties when using 4-fluorophenylboronic acid (which was superior to that of phenylboronic acid and 4-carboxyphenylboronic acid), this reagent was selected and used with other fluorinated bromoaryls, namely, 2-Bromofluorobenzene, 1-bromo-3-fluorobenzene, 2-bromo-5-fluorotoluene, and 2-bromo-4-fluorotoluene, for the formation of different difluorinated biphenyls. In the case of 2-Bromofluorobenzene, 1-bromo-3-fluorobenzene, and 1-bromo-4-fluorobenzene at both 70 °C or 110 °C and 3 h of reaction, the obtained TOF values were between ca. 44 and 67 h−1. In contrast, in the case of 2-bromo-5-fluorotoluene and 2-bromo-4-fluorotoluene at 70 °C or 110 °C and 3 h of reaction, the TOF values were lower, indicating an inferior activity when using this substituted bromide.[1]
In order to determine the influence of different fluorinated aryl bromides in the catalytic C-C coupling reaction, additional reactions of 1-bromo-3-fluorobenzene, 2-Bromofluorobenzene, 2-bromo-5-fluorotoluene, or 2-bromo-4-fluorotoluene with 4-fluorophenylboronic acid were carried out, following a similar procedure to using temperatures of 70 °C and 110 °C and reaction times of 3, 8, 24, or 48 h. In this case, the quantification was carried out by comparison of the ratio of the integral of the signal of the fluorine atom in the 19F NMR spectrum of 1-bromo-3-fluorobenzene (δ −106.9 ppm), 2-Bromofluorobenzene (δ −104.4 ppm), 2-bromo-5-fluorotoluene (δ −112.0 ppm), or 2-bromo-4-fluorotoluene (δ −112.0 ppm) with that of the standard (4-fluorobenzophenone, δ 102.9 ppm) before and after the reaction. For an example of the 19F NMR spectra of the reaction between 4-fluorophenylboronic acid and 2-Bromofluorobenzene. In this work we have synthesized a hybrid heterogeneous catalyst based on Pd-supported nanoparticles onto COOH-modified graphite support in a mixture of phases (G-COOH-Pd-10). In addition, the recyclability of the system G-COOH-Pd-10 at short 3 h reaction time shows a progressive loss of activity in the catalytic tests from 90% to 47% after the fifth cycle. However, this material showed a good degree of recyclability with a low deactivation of less than 8% after up to five catalytic cycles after 48 hours, probably due to the longer reaction time compensating for the loss of activity.
Palladium-catalyzed aminocarbonylation/SNAr approach to dibenzoxazepinones
Seven-membered heterocycles are receiving continuing attention as their skeletons are widely present in numerous pharmaceuticals and natural products. Among these seven-membered heterocycles, dibenzo[b,e][1,4]oxazepin-11(5H)-one derivatives represent a class of versatile compounds owing to their promising pharmaceutical and biological activities, including HIV-1 RT inhibition, H4R agonist, antidepressant, anti-psychotic, anti-tumor, antioxidant and anti-inflammatory7 activities. Initially, 2-bromofluorobenzene and 2-aminophenol were selected as the model substrates to optimize the reaction conditions. A preliminary study was carried out on a 0.5 mmol scale at 120 °C in DMAc, using BuPAd2 as the ligand and DBU as the base, affording 3a in 75% isolated yield. With the optimized reaction conditions in hand, we further investigated the substrates’ scope of this procedure. Several substituted 2-bromofluorobenzenes were subjected to the optimized conditions described above. A convenient procedure for the synthesis of dibenzoxazepinones has been developed. Utilizing the protocol of one-pot palladium-catalyzed aminocarbonylation/aromatic nucleophilic substitution (SNAr) sequence, with 2-aminophenols and 2-bromofluorobenzenes as the substrates, the desired dibenzo[b,e][1,4]oxazepin-11(5H)-ones were prepared in moderate to excellent yields. The broad substrate scope and functional group tolerance of the reaction makes this approach a practical method for the synthesis of valuable dibenzoxazepinone and its derivatives. Mechanistic studies suggest that aminocarbonylation proceeds prior to SNAr.[2]
References
[1]Sadeghi Erami, R., Díaz-García, D., Prashar, S., Rodríguez-Diéguez, A., Fajardo, M., Amirnasr, M., & Gómez-Ruiz, S. (2017). Suzuki-Miyaura C-C Coupling Reactions Catalyzed by Supported Pd Nanoparticles for the Preparation of Fluorinated Biphenyl Derivatives. Catalysts, 7(3), 76.
[2]Shen, C., Neumann, H., & Wu, X.-F. (2015). A highly-efficient palladium-catalyzed aminocarbonylation/SₙAr approach to dibenzoxazepinones. Green Chemistry, 17(5).
You may like
Lastest Price from 2-Bromofluorobenzene manufacturers
US $0.00/kg2025-11-25
- CAS:
- 1072-85-1
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- Customise

US $10.00/KG2025-04-21
- CAS:
- 1072-85-1
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt