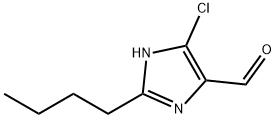
2-Butyl-4-chloro-5-formylimidazole as Pharmaceutical Intermediacy
2-Butyl-4-chloro-5-formylimidazole is an API intermediate used as an intermediate for the anti-hypertensive drug Chlosartan. It can be used in the chemical manufacture of many APIs and advanced intermediates. This substance typically appears as solid flakes or fine powder at ambient temperature. It comes in crystalline form, showing a color from off-white to faint yellow. In pharmaceutical research, 2-Butyl-4-chloro-5-formylimidazole acts as a building block for active pharmaceutical ingredient synthesis, notably for drugs that require an imidazole-based core altered by selective halogenation or aldehyde functionalization. Its robust reactivity profile makes it popular during step-growth synthesis where specificity and minimal side reactions matter.

Novel Schiff Bases Derived from 2-Butyl-4-chloro-5-formylimidazole
Imidazole and its derivatives are considered vital class of heterocyclic compounds that exist in different natural products and biological systems. They play a vital role in medicine due to their viable property and have a wide range of applications such as antibacterial, antihypertensive, antifungal, anti-inflammatory, tubercular, antiviral, anticonvulsant, and anthelmintic. Hence, the antimicrobial study of substituted imidazole’s such as 2-butyl-4-chloroformyl imidazole’s is very interesting to develop new drugs for addressing the challenges in the medicinal world. The Schiff base ligands, BN-21 and BN-22, have been synthesized by reacting 2-butyl-4-chloroimidazole with thiosemicarbazone, BN-49 2-Butyl-4-chloro-5-formylimidazole, and benzophenone hydrazone, whereas BN-16 was obtained from carbazolone and benzophenone hydrazone. The significance of this study is to synthesize the Schiff base ligands using carbonyl compounds and semi-carbazide or hydrazone with alcoholic solvent as reaction media in the presence of an acid catalyst followed by characterization using UV–Visible, IR, and high-resolution mass and NMR spectra studies to confirm their chemical structure. The novel Schiff base ligands were designed as potential candidates for anti-hypertensive and anti-emetic drugs, with 2-Butyl-4-chloro-5-formylimidazole as the key component of the molecules for bioactivity, and similarly, tetrahydro-N-methyl carbazolone played a key role in inducing bioactivity in synthesized molecules. The designed ligands were synthesized in the presence of an acid catalyst by refluxing carbonyl compounds and semi-carbazide or hydrazone with alcoholic solvent as reaction media.[1]
In this study, a novel Schiff base was synthesized which comprises a core moiety of 2-Butyl-4-chloro-5-formylimidazole. The ligand was synthesized by the reaction between the carbonyl compound 4-[(2-butyl-4-chloro-5-formyl-1H-imidazol-1-yl) methyl] benzoate and primary hydrazine compounds such as 2,4-dinitrophenylhydrazine in the presence of an alcoholic solvent and an acid catalyst. The synthesized Schiff base ligand is characterized by mass and spectral analysis including NMR. The appearance of extended conjugation of the π-electrons system between active 2-Butyl-4-chloro-5-formylimidazole moieties with nitro substituted phenyl ring. The ligands are assessed for an antibacterial activity for Escherichia coli and Staphylococcus aureus to evaluate the inhibition potential by MIC and well diffusion method. The biological activity of the ligand has shown a significant property against the Gram-negative bacterium, E. coli, and Gram-positive bacterium, S. aureus of about 27 mm and 28 mm of inhibitory action, respectively. This study paves the way for the development of novel antimicrobial agents for emerging clinical pathogens.
Design of 2-Butyl-4-chloro-5-formylimidazole-derived 1,3,4-oxadiazoles
A novel series of eight compounds, 2-Butyl-4-chloro-5-formylimidazole-derived 1,3,4-oxadiazoles, were designed and synthesized conveniently over four steps in good yield. The synthesized 1,3,4-oxadiazoles were confirmed via various analytical techniques, such as FTIR, 1H NMR, 13C NMR, and mass spectrometry. The ACE enzyme inhibitory potential of the synthesized compounds was evaluated, with compounds 5b and 5c showing IC50 values of 81.29 ± 0.70 and 51.01± 1.58 µM, respectively. Additionally, the synthesized compounds were screened for anticancer activity against the MDA-MB-231 and Caco-2 cell lines, and an antitubercular evaluation against the M. tuberculosis H37RV strain was also carried out. In silico studies, such as molecular docking to determine orientation and binding interactions, ADME prediction to determine Lipinski's rule, and molecular dynamics simulations to evaluate ligand‒protein stability were also conducted.[2]
References
[1]Pandiarajan S, Hajarabeevi N, Shaikh RR, Raghunathan S, MubarakAli D. Synthesis and Characterization of Novel Schiff Bases Derived from 2-Butyl-4-chloro Imidazole for the Enhanced Antimicrobial Property. Appl Biochem Biotechnol. 2023 Jan;195(1):253-263. doi: 10.1007/s12010-022-04112-2. Epub 2022 Sep 7. PMID: 36070167.
[2]R, M., Anantharaju, P. G., Madhunapantulas, S. V., & Gaonkar, S. L. (2025). Design, synthesis, and biological evaluation of 2-butyl-4-chloroimidazole-derived 1,3,4-oxadiazoles: ACE inhibition, anticancer, and antitubercular activities. Journal of Molecular Structure, 1322(Part 4), 140630.
You may like
See also
Lastest Price from 2-Butyl-4-chloro-5-formylimidazole manufacturers

US $1.00/KG2025-04-21
- CAS:
- 83857-96-9
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt

US $0.00-0.00/kg2025-04-04
- CAS:
- 83857-96-9
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- 1Ton