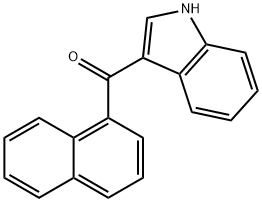
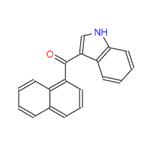
3-(1-Naphthoyl)indole: Properties, Detection & Pharmacology
3-(1-Naphthoyl)indole is a synthetic cannabinoid that is structurally similar to THC. It has been detected in human urine, plasma and autopsy samples by high-resolution mass spectrometry. As a key chemical and pharmaceutical intermediate, it can be synthesised via a Friedel-Crafts acylation reaction between indole and 1-naphthylformaldehyde reagent, finding extensive application in organic compound synthesis and pharmaceutical research. Additionally, 3-(1-Naphthoyl)indole belongs to the class of synthetic cannabinoids, sharing structural similarities with tetrahydrocannabinol (THC). Possessing certain psychoactive properties and inherent hazards, it must be stored in a sealed container at 2–8°C in a dry environment. When handling, appropriate protective measures should be taken to avoid direct contact with skin or eyes.

Monoclonal Antibodies Specific for 3-(1-naphthoyl) Indole Derivatives
Synthetic cannabinoids have attracted much public attention recently in Japan. 3-(1-Naphthoyl)indole derivatives, such as 1-butyl-3-(1-naphthoyl) indole (JWH-073) and 1-pentyl-3-(1-naphthoyl) indole (JWH-018), are known as synthetic cannabinoids and are also considered dangerous illegal drugs in Japan. It has become necessary to develop sensitive and useful methods for detection of 3-(1-naphthoyl) indole derivatives such as JWH-018 and JWH-073. Methods such as gas chromatography/mass spectrometry (GC/MS) and liquid chromatography/mass spectrometry (LC/MS) are possible to apply for the detection of 3 3-(1-Naphthoyl)indole derivatives. But these detection methods are difficult to use and are generally time consuming. There are various types of immunological assays used as qualitative and quantitative methods for the detection of other drugs such as methamphetamine and cocaine, using monoclonal antibodies against these drugs. Development of a specific immunological detection method for 3-(1-naphthoyl) indole derivatives requires a monoclonal antibody specific for 3-(1-naphthoyl) indole derivatives. Here we describe the production and characterization of new monoclonal antibodies specific for 3-(1-Naphthoyl)indole derivatives.[1]
1-(heptyl-7-carboxylate)-3-(1-naphtoyl) indole and 1-(heptyl-7-carboxylate)-3-(1-naphtoyl) indole-KLH conjugate were synthesized for use as an immunogen to prepare an antibody specific for 3-(1-Naphthoyl)indole derivatives. Approximately 3.6 molecules of 1-(heptyl-7-carboxylate)-3-(1-naphtoyl) indole derivative were introduced per 1 mol of KLH. Mouse splenocytes produced a high-titer antibody immune response to the immunogen and were fused with myeloma cells resulting in hybridomas that were screened for the secretion of anti-3-(1-naphtoyl) indole derivative-specific antibodies by non-competitive ELISA. Both of the antibodies (NT01, IgG1; NT02, IgG1) were found to be specific for 3-(1-naphtoyl) indole derivatives such as 1-methyl-3-(1-naphthoyl) indole, 1-ethyl-3-(1-naphthoyl) indole, and 1-octyl-3-(1-naphthoyl) indole using competitive ELISA. However they failed to bind to indole, naphtoic acid, and 4-methyl-naphtoic acid. The 3-(1-Naphthoyl)indole structure appears necessary for antibody recognition. In addition, the MAbs that were produced from the NT01 hybridoma has a high affinity for all of the 3-(1-naphthoyl) indole derivatives described above (IC50 = 60 – 100 nM). JWH-018 and JWH-073, which are well known as illegal drugs, were also one of the 3-(1-naphthoyl) indole derivatives. The length of the alkyl chain is only different from JWH-018 and 1-ethyl-3-(1-Naphthoyl)indole. The two MAbs that were produced in this study will certainly be bound to JWH-018 and JWH-073. In future studies, the anti-3-(1-naphthoyl) indole derivative MAbs that were obtained in this study will be used as an immunosensor for illegal drugs and evaluated on real samples.
Synthesis and Pharmacology of 1-Alkyl-3-(1-naphthoyl)indoles
To develop SAR at both the cannabinoid CB1 and CB2 receptors for 3-(1-naphthoyl)indoles bearing moderately electron withdrawing substituents at C-4 of the naphthoyl moiety, 1-propyl and 1-pentyl-3-(4-fluoro, chloro, bromo and iodo-1-naphthoyl) derivatives were prepared. To study the steric and electronic effects of substituents at the 8-position of the naphthoyl group, the 3-(4-chloro, bromo and iodo-1-naphthoyl)indoles were also synthesized. The affinities of both groups of compounds for the CB1 and CB2 receptors were determined and several of them were evaluated in vivo in the mouse. The effects of these substituents on receptor affinities and in vivo activity are discussed and structure-activity relationships are presented. Although many of these compounds are selective for the CB2 receptor, only three JWH-423, 1-propyl-3-(4-iodo-1-naphthoyl)indole, JWH-422, 2-methyl-1-propyl-3-(4-iodo-1-naphthoyl)indole, the 2-methyl analog of JWH-423 and JWH-417, 1-pentyl-3-(8-iodo-1-naphthoyl)indole, possess the desirable combination of low CB1 affinity and good CB2 affinity. Together, these results suggest that steric effects play a far more prominent role in determining the nature of the interaction of this series of 1-alkyl-3-(1-naphthoyl)indoles with the identified cannabinoid receptors than do electronic effects.
References
[1]Nakayama, Hiroshi et al. “Development and Characterization of Monoclonal Antibodies Specific for 3-(1-naphthoyl) Indole Derivatives.” Monoclonal antibodies in immunodiagnosis and immunotherapy vol. 35,1 (2016): 48-51. doi:10.1089/mab.2015.0045
[2]Wiley JL, Smith VJ, Chen J, Martin BR, Huffman JW. Synthesis and pharmacology of 1-alkyl-3-(1-naphthoyl)indoles: steric and electronic effects of 4- and 8-halogenated naphthoyl substituents. Bioorg Med Chem. 2012 Mar 15;20(6):2067-81. doi: 10.1016/j.bmc.2012.01.038. Epub 2012 Jan 30. PMID: 22341572; PMCID: PMC3298571.
You may like
US $0.00-0.00/kG2025-09-24
- CAS:
- 109555-87-5
- Min. Order:
- 1kG
- Purity:
- 99%
- Supply Ability:
- 1000KG

US $0.00/kg2025-03-18
- CAS:
- 109555-87-5
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 10T