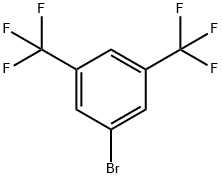
3,5-Bis(trifluoromethyl)bromobenzene: Synthesis of Fluorosilane Monomers
3,5-Bis(trifluoromethyl)bromobenzene is a significant member of fluorinated aromatic compounds. Its molecular structure centres on a benzene ring, with two strongly electron-withdrawing trifluoromethyl groups (-CF₃) symmetrically substituted at the meta position. A highly reactive bromine atom is bonded at the para position. The strong electron-withdrawing groups significantly enhance the reactivity of the bromine atom, making ,5-Bis(trifluoromethyl)bromobenzene an excellent arylating intermediate and nucleophilic substitution substrate in organic synthesis.

Synthesis of Trifluoromethylphenyl Silane Monomers using 3,5-Bis(trifluoromethyl)bromobenzene
Fluorosilicone materials play a great role in the field of polymer material, because they possess the advantages of organosilicone and organofluorine polymers. Recently, the study of fluorosiloxane polymers has been focused on the polysiloxanes with long-chain per-fluorine group substitution. This provided the fluoro-polysiloxanes not only the excellent anti-solvent, thermal stability, with weathering and wear-resistance performances, but especially the surface energy as low as7.0 mN/m. 1 The resulting fluorosilicone oil has unique performance when it is used as surface modification material, and has very broad application prospects in fabric finishing, floor polishes, paints, coating additives,defoamers2 and the hydraulic fluid of aviation industry. 3,5-Bis(trifluoromethyl)bromobenzene was pur- chased from Beijing Golden Olive Company and used without any purification,whose purity was above 98%. Ether and tetrahydrofuran (THF)were both dried by reflux with sodium and then distilled.[1]
3,5-Bis(trifluoromethyl)phenyl(methyl)dimethoxysilane: Magnesium (2.88 g,0.12 mol)and ether were placed in a 500 mL round-bottomed three-necked flask equipped with a magnetic stirrer bar, a thermometer, a pressure equalizing dropping funnel and a con- denser. The mixture was stirred and heated to 30 C un- der nitrogen, and a solution of 3,5-Bis(trifluoromethyl)bromobenzene (29.3 g,0.10 mol)in ether was added dropwise to the stirred mixture. After the addition was complete, the reaction mixture was stirred at 30 C for another 2 h, then cooled to room temperature and added dropwise to a solution of methyltrimethoxysilane(55.2 g,0.40 mol)while maintaining the temperature below 5 C. After 2 h the addition was complete and the resultant reaction mixture was stirred below 5 C for 1 h and then filtered, the filtrate was concentrated in vacuo to remove ether and un-reacted methyltrimethoxysilane, diluted with hexane and filtered through a filter paper. The resultant filtrate was concentrated in vacuo to obtain a brown liquid, which was distilled to give the title product (16.8 g) as a clear liquid. The yield was 56.3% (GC purity being 97.9%). Bis[3,5-bis(trifluoromethyl)phenyl](methyl)- methoxysilane was synthesized according to the procedure similar to that described for 1a,the Grignard reaction was initiated between magnesium (5.4 g, 0.23 mol)and 3,5-bis(trifluoromethyl)bromobenzene(43.95 g,0.15 mol)in ether. Then the Grignard reagent was added to 10.35 g(0.08 mol)of methyltrimethoxy- silane at 60 C. Stirring was continued for 3 h. By the similar treatment procedure 2a was obtained with the yield being 21.2%,and the purity being 95.3%.
Tri[3,5-bis(trifluoromethyl)phenyl]methylsilane: Grignard reagent was prepared by 1.73 g (0.07 mol)of magnesium and 17.58 g (0.06 mol)of 3,5-bis(trifluoromethyl)bromobenzenein THF. Then 2.74 g(0.02 mol)of methyltrimethoxysilane was added drop-by-drop to the stirred reaction mixture, and then the mixture was refluxed with stirring for 3 h. Subsequently the liquid portion of the reaction product was poured into a mixture of ice and ammonium chloride saturated water solution. After extraction with hexane, the organic layer was collected, dried with Na2SO4 and concentrated in vacuo to obtain a brown liquid. After cooling to room temperature, crude solid product was separated out, which was ground in anhydrous MeOH to give white solid 3a.The yield was 30.3%.The synthesis of tri[3,5-bis(trifluoromethyl)- phenyl]ethoxylsilane was performed using 5.4 g of magnesium (0.23 mol),43.95 g of ,5-Bis(trifluoromethyl)bromobenzene (0.15 mol)and 10.4 g of ethyl silicate (0.05 mol)according to the synthesis procedure of 3a in THF. The Grignard reagent was added to ethyl silicate at 60 C, and the mixture was refluxed with stir- ring for 3 h. After the solvent was removed, the residue was diluted with hexane and filtered, the resultant filtrate was concentrated in vacuo, then the brown liquid was cooled to room temperature, and crude solid product was separated, which was added with anhydrous methanol. The white solid product of 3b was separated out by filtration in the yield 24.7%.
References
[1] Zhanxiong Li. (2010). Synthesis and Characterization of Novel Trifluoromethylphenyl Silane Monomers. Chinese Journal of Chemistry, 27 12, 2429–2432.
See also
Lastest Price from 3,5-Bis(trifluoromethyl)bromobenzene manufacturers
US $0.00-0.00/KG2025-07-05
- CAS:
- 328-70-1
- Min. Order:
- 1KG
- Purity:
- 99.0%
- Supply Ability:
- 10000KGS

US $0.00/KG2025-04-21
- CAS:
- 328-70-1
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 30tons/month