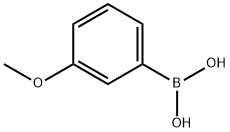
3-Methoxyphenylboronic Acid in Organic Synthesis
3-Methoxyphenylboronic acid is a commonly used arylboronic acid compound featuring a methoxy-substituted benzene ring and a boronic acid functional group, exhibiting excellent stability and reactivity. This compound is widely employed in Suzuki–Miyaura coupling reactions as a key intermediate for constructing diaryl structures, holding significant value in the synthesis of pharmaceutical molecules, pesticides, and organic functional materials.

Synthesis of 3,6-diaryl-1H-pyrazolo[3,4-b]pyridines
Arylboronic acids, such as 3-Methoxyphenylboronic acid, play a central role in modern cross-coupling chemistry due to their stability, functional-group tolerance, and broad applicability in C–C bond-forming reactions. Among these, transition-metal-catalysed cross-coupling reactions have been well-established as powerful synthetic tools for constructing complex aromatic frameworks. The transition-metal-catalysed cross-coupling reaction has been well-established as a powerful synthetic tool in C–C bond formation. In particular, the Suzuki–Miyaura cross-coupling reaction has shown widespread applications in natural product synthesis, the synthesis of medicinally important pharmacophores and organic materials.1 To explore further synthetic applications of transition-metal-catalyzed cross-coupling reactions, organocatalytic reactions and arylation of heteroaromatics have attracted attention in recent years. For substrate scope, coupling with 4-fluorophenylboronic acid facilitates excellent reaction yield (97%), when compared to coupling with electron-donating 4-methoxyphenylboronic acid resulted in 81% yield. The difluorophenyl group containing pyrazolopyridine coupled with 3-methoxyphenylboronic acid produced 4g in 63% yield. The present strategy is efficient enough to couple with heteroaromatic boronic acids producing diarylated product in 62–68% yield. [1]
In addition, we successfully arylated the pyrazolo[3,4-b]pyridine with benzo[d][1,3]dioxol-5-ylboronic acid, giving 57% of 4k. The 3-hydroxyphenylboronic acid was quite stable under these conditions, as the coupled product was isolated in 52% yield. The one-pot coupling reaction afforded diaryl-1H-pyrazolo[3,4-b]pyridines in moderate to good yields (43–72%).In a one-pot reaction diphenyl-1H-pyrazolo[3,4-b]pyridine was isolated in 72% yield. Insertion of 4-methoxyphenylboronic acid as a second arylating agent was successful, as 4b was isolated in 60% yield in 4h. The combination of 4-methylphenylboronic acid as the first arylating agent and 4-cyanophenylboronic acid as the second proved to be successful, with an isolated yield of 60%. The structure of 4f was determined by X-ray crystallography. Further, when 3-methoxyphenylboronic acid reacted with 6-chloro-3-(2,4-difluorophenyl)-1-(4-methoxybenzyl)-1H-pyrazolo[3,4-b]pyridine, the product was isolated in 48% yield in 6 h. We have observed that C3 arylation was preferred over C6 arylation, in our reaction conditions. But, we cannot neglect the role of I vs. Cl as one of the governing factors. Thus the one-pot method can be used as a time and cost-effective method for the diarylation of pyrazolopyridines.
Flow Suzuki Coupling with 3-Methoxyphenylboronic acid
Nucleosides are privileged structural motifs with a wide array of biological activity, that has led to significant investment in their syntheses over the past few decades. The current Covid-19 pandemic and the potential of nucleosides as therapeutics have further enhanced their appeal. The correct choice of solvent was necessary to maintain the homogeneity of the reaction mixture as any solid particle formation during the reaction could be detrimental to the flow system leading to channel blockage. The use of H2O:EtOH (2:1) as a solvent system allowed the catalytic transformation between I-dU (1a) and 3-methoxyphenylboronic acid (2a) with SerrKap palladacycle in the conventional Schlenck conditions under inert N2 atmosphere without any solid formation and was therefore applicable to continuous flow as the catalytic solution was observed to be completely homogeneous throughout the reaction. The product 3a was isolated (84%) upon column chromatography. Second, the screening of various catalytic conditions for the Suzuki-Miyaura cross-coupling of I-dU with 3-methoxyphenylboronic acid indicated SerrKap to be the best choice of catalyst. For the flow set-up, I-dU, SerrKap palladacycle and base (K3PO4) were dissolved in H2O:EtOH (3:2) and pumped via injector 1, while injector 2 was used for 3-methoxyphenylboronic acid in H2O:EtOH (3:2). The temperature of the reactor block was maintained at 60 °C (improved solubility of the substrate observed at 60 °C). Both solutions were pumped using an air-pump without the use of nitrogen gas.[2]
All the previous reactions were performed using 1.6 equiv. of 3-methoxyphenylboronic acid with respect to the starting I-dU. It was observed that the reduction in the amount of the boronic acid to 1.3 equiv. did not affect the catalytic efficiency, but any further reduction (1.2 eq.) led to a lower yield of the product. Base concentration (K3PO4) was found to affect the catalytic reactivity positively with 1.05 eq. providing higher product yield. To investigate the feasibility of coupling I-dC with 3-methoxyphenylboronic acid, reaction screening studies were conducted. After a thorough investigation that would promote the coupling to proceed in a relatively faster reaction time, not requiring the use of inert nitrogen atmosphere and avoiding chromatographic isolation of the product, the optimum conditions were obtained with 1.0 mol% SerrKap catalyst at a flow rate of 0.6 mL/min (H2O:EtOH, 12 + 8 mL) and pressure of 8–9 kPa, which provided best yield of 90% in 29 minutes. The application of flow catalytic conditions has once again improved the reactivity and drastically reduced the reaction time and allowed the reaction to be conducted in air rather than maintaining inert conditions.
References
[1]Tandon, V. et al. Synthesis of 3,6-diaryl-1H-pyrazolo[3,4-b]pyridines via one-pot sequential coupling… RSC Advances (2018). Article: C8RA07104G.
[2]Gaware S, Kori S, Serrano JL, Dandela R, Hilton S, Sanghvi YS, Kapdi AR. Rapid plugged flow synthesis of nucleoside analogues via Suzuki-Miyaura coupling and heck Alkenylation of 5-Iodo-2'-deoxyuridine (or cytidine). J Flow Chem. 2023 Mar 16:1-18. doi: 10.1007/s41981-023-00265-1. Epub ahead of print. PMID: 37359287; PMCID: PMC10019434.
You may like
Lastest Price from 3-Methoxyphenylboronic acid manufacturers

US $6.00/kg2025-04-21
- CAS:
- 10365-98-7
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 2000KG/Month

US $7.00/KG2025-03-28
- CAS:
- 10365-98-7
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 20TONS