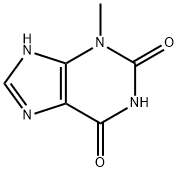
3-Methylxanthine: Gut-Derived Metabolite
3-methylxanthine is a metabolite produced during the degradation of theophylline by certain microorganisms, such as Aspergillus tamarii and Aspergillus ustus, during the microbial fermentation of dark tea. It is involved in the bioconversion processes that modify chemical contents in dark tea.

Gut-derived metabolite 3-methylxanthine enhances cisplatin-induced apoptosis
The gut microbiota has garnered increasing attention due to its influence on the development of anticancer responses. Study have shown that Bacteroides fragilis is essential for the immunostimulatory effect of cytotoxic T-lymphocyte antigen 4(CTLA-4) blockade. In this study, we present evidence demonstrating that the depletion of gut microbiota induced by ABXs exacerbates the anticancer efficacy of cisplatin in mice with OC. Metabolomic analysis revealed a significant elevation of 3-methylxanthine in ABX-treated mice. Our findings indicated that metabolites exhibiting structural similarity with it interact with genes enriched in cancer-related pathways. Moreover, we identified that 3-methylxanthine enhances cisplatin-induced apoptosis both in vivo and in vitro. Mechanistically, through comprehensive multiomic analysis, we discovered a positive correlation between 3-methylxanthine and dopamine receptor D1 (DRD1). Subsequently, we demonstrated that both small interfering RNA (siRNA) -targeting DRD1 and pharmacological inhibitors effectively attenuate the augmented cisplatin-induced apoptosis provoked by 3-methylxanthine. These results provide preliminary insights into the mechanism, by which modulates the sensitivity of cisplatin in OC cells.[1]
The precise correlation between the gut microbiota and the anticancer effect of cisplatin in OC remains inadequately understood. Our investigation has revealed that manipulation of the gut microbiota via the administration of antibiotics amplifies the efficacy of cisplatin through the facilitation of apoptosis in OC-bearing mice. Metabolomic analysis has demonstrated that the cecum content from antibiotic-treated mice exhibits an increase in the levels of 3-methylxanthine, which has been shown to potentially enhance the therapeutic effectiveness of cisplatin by an integrated multiomic analysis. This enhancement appears to be attributable to the promotion of cisplatin-induced apoptosis, with 3-methylxanthine potentially exerting its influence via the dopamine receptor D1-dependent pathway. These findings significantly contribute to our comprehension of the impact of the gut microbiota on the anticancer therapy in OC. Notably, the involvement of 3-methylxanthine suggests its prospective utility as a supplementary component for augmenting treatment outcomes in patients afflicted with ovarian cancer.
OC patients initially exhibit a favorable response to surgical debulking and platinum-based chemotherapy. However, relapse with platinum-resistant cancer frequently occurs, and the effectiveness of novel immunotherapy in OC is limited, with a response rate below 10%. Through comprehensive multiomic studies, we have identified a key gut-derived metabolite, 3-methylxanthine, as a significant regulator in modulating the response to cisplatin. Notably, our further investigations have confirmed that 3-methylxanthine enhanced cisplatin-induced apoptosis through the DRD1 pathway. These findings underscore the potential significance of a “gut–ovary” axis in regulating the effectiveness of anticancer therapies, with gut-derived metabolites demonstrating promise as safe and promising small molecule compounds for enhancing the sensitivity of cisplatin. In conclusion, our study presents compelling evidence of a cross-talk mechanism between gut microbiota and the anticancer efficacy of cisplatin in an OC mouse model. Furthermore, we have identified a key gut-derived metabolite, 3-methylxanthine, which enhances the response to cisplatin by promoting apoptosis through DRD1 activation. This discovery sheds light on the remarkable role of the gut-ovary axis in regulating anticancer therapy and unveils potential therapeutic avenues for targeting cancer. The combination of cisplatin with gut-derived metabolites as adjuvants holds promising translational value in the context of anticancer therapy.
Progress in Methylxanthine Biosynthesis
In monomethyl xanthine, 3-methylxanthine (3-MX) is a prominent example. It is characterized in that the N-3 position of the purine ring is connected with methyl, which makes it a methylated derivative of xanthine. Functionally, 3-methylxanthine has various pharmacological effects. It can effectively stimulate the central nervous system and improve an individual’s alertness and attention level. This effect may be attributed mainly to its inhibitory effect on adenosine receptors. This mechanism helps reduce the inhibitory effect of adenosine on neurons, thus enhancing the transmission of nerve signals and brain activity. Additionally, 3-methylxanthine can influence lipid metabolism and energy expenditure, with moderate consumption potentially aiding in weight management. Algharrawi et al. utilized the ndmA and ndmD genes from Pseudomonas putida to engineer an E. coli strain capable of producing 3-MX from theobromine. In a study by Mani Subramanian et al., increasing the copy number of the ndmA and ndmD genes in E. coli contributed to the high-yield production of 3-MX from theobromine.[2]
In a study by Chang Liu et al., high yields of 7-MX were achieved during the development of their production process, prompting them to also explore the synthesis of 3-methylxanthine. Owing to the very low solubility of theobromine, which makes it unsuitable for production via large-scale fermentation, they chose to use theophylline as the substrate for 3-MX production. By utilizing the ndmA and ndmD enzymes, the engineered strain was able to convert theophylline to 3-methylxanthine, achieving a final yield of 22.96 ± 0.81 mM. This represents the highest known biosynthetic yield of 3-MX to date. The exploration of their applications continues to deepen, with these derivatives being utilized in the pharmaceutical industry for the development of novel drugs and adjunctive therapies to address pressing challenges such as cardiovascular diseases, diabetes, and neurodegenerative disorders. In the cosmetics industry, their antioxidant properties have led to the creation of innovative antiaging and skin-whitening products.
References
[1]Mai Z, Han Y, Liang D, Mai F, Zheng H, Li P, Li Y, Ma C, Chen Y, Li W, Zhang S, Feng Y, Chen X, Wang Y. Gut-derived metabolite 3-methylxanthine enhances cisplatin-induced apoptosis via dopamine receptor D1 in a mouse model of ovarian cancer. mSystems. 2024 Jul 23;9(7):e0130123. doi: 10.1128/msystems.01301-23. Epub 2024 Jun 20. PMID: 38899930; PMCID: PMC11264688.
[2]Jiang T, Zuo S, Liu C, Xing W, Wang P. Progress in Methylxanthine Biosynthesis: Insights into Pathways and Engineering Strategies. Int J Mol Sci. 2025 Feb 11;26(4):1510. doi: 10.3390/ijms26041510. PMID: 40003976; PMCID: PMC11855574.
You may like
See also
Lastest Price from 3-Methylxanthine manufacturers

US $0.00-0.00/Kg/Drum2025-04-21
- CAS:
- 1076-22-8
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 500kg

US $100.00-60.00/kg2025-03-07
- CAS:
- 1076-22-8
- Min. Order:
- 100kg
- Purity:
- 99.99%
- Supply Ability:
- 100Tons