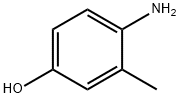
4-Amino-3-methylphenol: Synthesis & Analytical Determination
4-Amino-3-methylphenol, which according to the web-based International Cosmetic Ingredient Dictionary and Handbook (wINCI; Dictionary) is reported to function in cosmetics as a hair colorant, was previously reviewed by the Expert Panel for Cosmetic Ingredient Safety (Panel) as part of a safety assessment of 6 amino-cresol hair dye ingredients that was published in 2004. At that time, the Panel concluded that “the available data support the safety of 4-Amino-3-methylphenol for use in oxidative and nonoxidative (semi-permanent) hair dyes.” In accordance with its Procedures, the Panel evaluates the conclusions of previously-issued reports approximately every 15 years, and it has been at least 15 years since this assessment has been issued. In June 2022, the Panel determined that this safety assessment should be re-opened for re-evaluation due to several of the other amino-cresol hair dye ingredients that were included in the original 2004 report being banned for use in cosmetics by the European Commission. However, because the Panel determined that data for these amino-cresol hair dye ingredients could not be read-across, rather than including all 6 ingredients in one amended report, re-reviews of each hair dye will be presented as individual stand-alone reports.

Preparation method for 4-amino-3-methylphenol
4-amino-3-methylphenol is Material of Fluoran dyestuff, plant protection product and the synthetic necessary intermediate of medicine, is industrial important raw and processed materials; Now high-quality 4-amino-3-methylphenol is also for the preparation of the synthetic and carbon fiber of high-end resin. The first kind is to be raw material with Ortho Nitro Toluene, by reducing and having reset reaction, has following the whole bag of tricks: (a) electrochemical method, and this method negative electrode adopts poisonous mercury salt, and productive rate is low; (b) aluminium-aqueous solutions of organic acids, this method products obtained therefrom purity is low, and cost is high; (c) in dilute sulphuric acid, with hydrogen, carry out the method for catalytic reduction, this method catalyzer adopts the precious metals such as platinum, rhodium, palladium; Long reaction time, productive rate is low. The preparation method who the invention provides a kind of new high-quality 4-amino-3-methylphenol reduces environmental pollution in the quality of improving the quality of products, and makes that products production cleans, environmental protection, makes product production in enormous quantities preferably.[1]
In 2000L stainless steel autoclave, suck methyl alcohol 1000kg, pass into liquefied ammonia 5kg, 4-nitroso-group-3-methylphenol 240kg, drop into 5% palladium carbon 2kg, envelope still, nitrogen replacement 3 times, hydrogen exchange 3 times, reactor hydrogen pressure keeps 0.1~0.2Mpa, then be warming up to 20~25 ℃, start reaction, no longer change to hydrogen pressure, reaction 8h, sampling detects to 4-nitroso-group-3-monomethylaniline and reacts completely.Pressure release, reaction solution is removed to catalyzer by strainer, decompression steams methyl alcohol, steam about 600kg to methyl alcohol, be cooled to 10 ℃, be discharged in filtering jar, then centrifugal 4-amino-3-methylphenol crude product that obtains, 4-amino-3-methylphenol the crude product obtaining is put in the methanol aqueous solution 600kg of 50% concentration again, be warming up to 45 ℃, insulated and stirred 20min, then be cooled to 5~10 ℃, centrifugal, 45 ℃ of fast boilings are dried and obtain 4-amino-3-methylphenol highly finished product 99.4kg, and yield is 80.5%, product HPLC >=99.7%.
Determination of 4-Amino-3-methylphenol using fluorescamine
Since ancient times people have the desire to change their hair colour. In earlier times especially natural hair dyes, e.g. henna or indigo were used, whereas since the 1950s synthetic hair dyes have become more and more important. Generally, there are three basic categories of hair dyes: temporary, semi-permanent and permanent hair dyes. Permanent hair dyes, the most popular group, can be sub-divided into oxidation hair dyes and progressive hair dyes. The purpose was to improve the limit of detection (LOD) by fluorescence detection compared to analysis in the UV range. Further investigations were carried out to develop an optimized solid-phase extraction method for the determination of 4-Amino-3-methylphenol and 5-amino-o-cresol in aqueous solution which can be used for excised pig skin model used for the estimation of dermal absorption in penetration studies as well. The derivatization reagent was prepared by dissolving 1 mg of fluorescamine in 5 ml acetonitrile. The derivatization reagent was stored at +4 °C in the refrigerator and was freshly prepared every day. Preparation of the derivatives was done by adding 200 μl of a 50 mM borate buffer pH 8.0 and 250 μl of the derivatization reagent to 50 μl of the stock solution of 4-Amino-3-methylphenol or 5-amino-o-cresol and following shaking on vortex. After a reaction time of 5 min at ambient temperature and shielded from light, 25 μl of the derivative were injected into the HPLC system.[2]
While 4-Amino-3-methylphenol shows highest UV absorbance at 240 nm, the maximum of 5-AC is at 220 nm because of the differing order of substituents. Analysis were performed on two different columns in order to obtain sharp peak form which is indispensable a precondition for detection as sensitive as possible. Mobile phase was for both substances ACN–water 5:95 (v/v). 4-Amino-3-methylphenol showed a retention time of 9 min on column B and a LOD of 10 ng/injection at a signal–noise ratio of 3:1, 5-AC was analyzed on column A having a retention time of 7.2 min and a LOD of 5 ng/injection. However, concentrations to be expected on principle in biological materials may be lower. Therefore, to improve the LOD for both compounds fluorescence derivatization was taken into consideration. Compared to detection in the UV range, LOD could be increased significantly. Despite similar molecular structure of the two investigated compounds, LOD of the fluorescamine derivatives differs extremely. Yet, concentrations in the ng- and pg-range can be determined without special effort. For the determination of 4-Amino-3-methylphenol and 5-amino-o-cresol in aqueous biological solutions as needed for penetration studies with the pig skin model, a solid-phase extraction procedure has been developed showing an average recovery of about 84%. The presented method for the determination of the 2 oxidation hair dyes 4-Amino-3-methylphenol and 5-amino-o-cresol is reproducible, simple and sensitive.
References
[1]CN103508908A
[2]Eggenreich, K., Zach, E., Beck, H., & Wintersteiger, R. (2004). Determination of 4-amino-m-cresol and 5-amino-o-cresol by high performance liquid chromatography and fluorescence derivatization using fluorescamine. Journal of Biochemical and Biophysical Methods, 61(1–2), 35–46.
You may like
See also
Lastest Price from 4-Amino-3-methylphenol manufacturers

US $10.00/KG2026-01-16
- CAS:
- 2835-99-6
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt

US $6.00/kg2025-04-21
- CAS:
- 2835-99-6
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 2000KG/Month