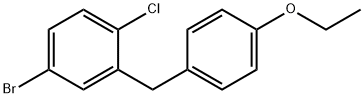
4-Bromo-1-chloro-2-(4-ethoxybenzyl)benzene: Building Block for Cross-Coupling
4-Bromo-1-chloro-2-(4-ethoxybenzyl)benzene is a substituted diphenylmethane derivative. Its molecular structure, featuring a bromo, chloro, and an ethoxybenzyl group, dictates its chemical reactivity and utility as a versatile building block in organic synthesis. The chemical behavior of 4-Bromo-1-chloro-2-(4-ethoxybenzyl)benzene is a direct consequence of its molecular architecture. The molecule consists of a central methylene bridge connecting a 4-ethoxyphenyl group to a 4-bromo-1-chlorophenyl group. The presence of two halogen atoms, bromine and chlorine, on one of the aromatic rings provides distinct sites for reactivity. The bromine atom is particularly susceptible to various metal-catalyzed cross-coupling reactions, such as Suzuki, Heck, and Sonogashira couplings. For optimal stability, 4-Bromo-1-chloro-2-(4-ethoxybenzyl)benzene should be stored at ambient or refrigerated (4°C) temperatures in a dry, well-sealed container. It should be kept away from strong oxidizing agents.

Synthesis of 4-Bromo-1-chloro-2-(4-ethoxybenzyl)benzene
Example 1: Et3SiH (6.76 mL, 42.34 mmol) was added to a solution of (5-bromo-2-chlorophenyl) - (4-ethoxyphenyl) methanone (6.0 g, 17.8 mmol) in 1:2 mixture of dry 1,2-dichloroethane/acetonitrile (2:1, 60 mL) and the mixture was cooled in an ice bath. BF3·OEt2 (2.53 mL, 20.51 mmol) was added and the mixture was heated at 50 °C for 3 h and then cooled at room temperature. Aqueous 7N KOH (30 mL) was added and the aqueous layer was extracted with dichloromethane (2 * 100 mL). The combined organic layers were washed with 2N KOH (2*) and brine and dried over Na2SO4. The solvent was removed under reduced pressure and the residue was chromatographed on a silica gel column (hexane/ethyl acetate 15:1) to give 4-Bromo-1-chloro-2-(4-ethoxybenzyl)benzene (5.6 g, 97%) as colorless crystals, with spectral and physical data identical to those reported in the literature, m.p. 40-42 °C (lit. m.p. 37 °C and 40-41 °C). 1H NMR (400 MHz, CDCl3) 7.20-7.28 (m, 3H), 7.08 (d, J = 8.8, 2H), 6.83 (d, J = 8.8, 2H), 4.00 (q, J = 7.0, 2H), 3.96 (s, 2H), 1.40 (t, J = 7.0, 3H); 13C NMR (125 MHz, CDCl3) 157.6, 141.3, 133.5, 133.1, 130.9, 130.5, 130.4, 130.0, 120.4, 114.6, 63.4, 38.2, 14.9. LC-MS (ESI positive) m/z: 325 [M+H]+.[1]
Example 2: Under nitrogen, 1 g of (5-bromo-2-chlorophenyl) - (4-ethoxyphenyl) methanone and10.88g dichloromethane was added to 100mL reaction flask, warmed to 30-40 ° C, added 2.88g acetonitrile, 0.22g sodium borohydride, stirred at 30-40 ° C for 0.5h, 0.96g trimethylchlorosilane was added dropwise, after dripping 30-40 for 3hrs, cooled to 0-5 , 0.63g of boron trifluoride diethyl ether was slowly added dropwise, the temperature was controlled <10 , after warming to 30-35 after dropping for 4hrs, cooled to 10-15 ° C, add 0.22g of sodium borohydride, 0.63g of boron trifluoride diethyl ether, the temperature was raised to 30-35 ° C reaction 4hrs, cooled to 0-5 ° C, 20mL methylene chloride was added slowly dropwise 20mL 12% sodium carbonate solution , Continue stirring after dropping 20mins, the layers were separated, the aqueous phase was extracted twice with 40mL of methylene chloride, the combined organic phases containing 4-Bromo-1-chloro-2-(4-ethoxybenzyl)benzene were washed once with water, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give 0.92g slightly yellow solid of 4-Bromo-1-chloro-2-(4-ethoxybenzyl)benzene, yield 96%.[2]
4-Bromo-1-chloro-2-(4-ethoxybenzyl)benzene applied in chemical synthesis
4-Bromo-1-chloro-2-(4-ethoxybenzyl)benzene can be used as a raw material for 4-(5-bromo-2~chloro-benzyl)-phenol: To a solution of 4-Bromo-1-chloro-2-(4-ethoxybenzyl)benzene (5.0 g, 15.35 mmol) in dichloromethane (20.0 mL) cooled to 0 degrees Celsius under nitrogen was added drop- wise over 30 minutes a 1 M solution of boron tribromide in dichloromethane (17.0 mL, 17.0 mmol). After the addition was complete, the reaction was allowed to warm to room temperature overnight (-16 hours). The reaction was cooled to 0 degrees Celsius and quenched with an aqueous solution of 1 N hydrochloric acid. The resulting mixture was stirred for 30 minutes and extracted using dichloromethane. The organic layer was separated and the aqueous layer was extracted two times with dichloromethane. The combined organic layers were dried over magnesium sulfate, filtered and concentrated under reduced pressure. The reaction was purified by flash chromatography over silica gel using the ISCO automated chromatography unit (120 g silica gel column) and eluting with a gradient of 0 to 100% ethyl acetate in heptane. 3.53 g of desired product obtained (77% yield). H NMR (400 MHz, METHANOL-^) delta ppm 3.94 (s, 2 H), 6.70 (d, J=8.6 Hz, 2 H), 6.98 (d, J=8.4 Hz, 2 H), 7.23 - 7.34 (m, 3 H).[3]
References
[1]Triantakonstanti, Virginia V.; Mountanea, Olga G.; Papoulidou, Kyriaki-Eleni C.; Andreou, Thanos; Koftis, Theocharis V.; Gallos, John K. Tetrahedron, 2018, vol. 74, # 39, p. 5700 - 5708
[2]CN106316803, 2017, A
[3]WO2011051864A1
You may like
Related articles And Qustion
See also
Lastest Price from 4-Bromo-1-chloro-2-(4-ethoxybenzyl)benzene manufacturers

US $1.00/g2025-08-11
- CAS:
- 461432-23-5
- Min. Order:
- 100g
- Purity:
- 99
- Supply Ability:
- 10000

US $0.00-0.00/kg2025-04-21
- CAS:
- 461432-23-5
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 1000kg