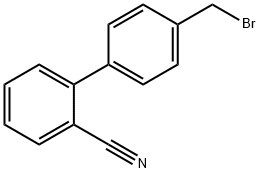
4-Bromomethyl-2-cyanobiphenyl: Thermodynamics & Green Photocatalytic Synthesis
4-Bromomethyl-2-cyanobiphenyl is an important pharmaceutical intermediate and organic synthetic material used in the preparation of drugs of the sartan class such as azilsartan ester and temisartan.

Thermodynamics of 4-Bromomethyl-2-cyanobiphenyl in different solvents
The compound 4-Bromomethyl-2-cyanobiphenyl is used as a synthetic intermediate for pharmaceutical products, such as losartan, valsartan and irbesartan. The sartans, as selective anqiotensin II receptor antagonist, are applied for antihypertensive, with the characteristics of long-lasting, good tolerance and high receptor-selectivity. Moreover, because of the outstanding clinical efficacy and good safety, the sartans have been developed rapidly in recent years. In pharmaceutical industry, high-purity 4-Bromomethyl-2-cyanobiphenyl is requisite for the production of the sartans. It is vital to separate and purify OTBNBr from the reaction solution by crystallization. In order to develop an optimal crystallization process, a deep understanding about the thermodynamics of the solid state OTBNBr and its solution is essential. Besides, investigations about the intermolecular interaction in OTBNBr solution are required to explain the differences of solubility in various solvents and it is also helpful for predicting the most appropriate solvents for its production. However, scant literature work about the thermodynamic properties of OTBNBr has been published. In this study, the melting properties of solid state 4-Bromomethyl-2-cyanobiphenyl were firstly investigated. The solubility of OTBNBr in various organic solvents was experimentally determined. COSMOthermX (COSMOlogic GmbH & Co. KG, Germany) was used to calculate the interaction energies between selected solvents molecules to provide a reasonable explanation about the striking differences of solubility in different solvents.[1]
Moreover, the van’t Hoff equation, the modified Apelblat equation, the λh equation and the Wilson model were used to correlate the measured solubility values. The activity coefficients of 4-Bromomethyl-2-cyanobiphenyl and the temperature dependence of van’t Hoff enthalpy in different solvents also were investigated and are discussed. In order to understand the thermodynamic behavior of OTBNBr in different solvents and to explain these solubility experimental results, COSMOthermX (COSMOlogic GmbH & Co. KG, Germany) was used to calculate the (solvent + solvent) intermolecular interaction energy in pure solvents. The total mean intermolecular interaction energy of investigated pure solvents from T = (283.15 to 323.15) K is presented. Obviously, the absolute values of intermolecular interaction energy of alcohols are much higher. This result suggests that the intermolecular interaction between organic solvent molecules plays an important role in the solubility of 4-Bromomethyl-2-cyanobiphenyl. It has been shown in the literature that the (solute + solvent) intermolecular interaction is closely related with the solubility of solute in solution. The melting properties of OTBNBr were experimentally determined.
The heat capacity difference between the pure solid state and the melt state 4-Bromomethyl-2-cyanobiphenyl was also experimentally determined. The enthalpy, entropy and Gibbs free energy of fusion were calculated by using the experimental values. The mole fraction solubility of OTBNBr in DMF, acetone, methyl acetate, ethyl acetate, butyl acetate, ethanol, n-butyl alcohol and isopropanol were measured over the temperature range from (283.15 to 323.15) K. The modified Apelblat equation, the λh equation, the Wilson model and the van’t Hoff equation were used to correlate the solubility data. It was found that the interaction of solvents molecules plays an important role in the solubility behavior of 4-Bromomethyl-2-cyanobiphenyl. From values of the activity coefficients obtained, it was also found that most selected solution systems deviate positively from Raoult’s law. Finally, based on a rigorous investigation, it was found that the assumption of temperature independence of the van’t Hoff enthalpy was not applicable to the 4-Bromomethyl-2-cyanobiphenyl solution systems tested.
Scale Light-Induced Synthesis of 4-Bromomethyl-2-cyanobiphenyl
The biphenyl motif has gained much interest in the research and synthesis of a diverse array of specialty compounds such as different classes of pharmaceuticals, luminescent sensors and photosensitizers. For pharmaceuticals, in particular, the 4-Bromomethyl-2-cyanobiphenyl intermediate has an important role in the commercial synthesis of the group of hypertensive drugs known as sartans. But it has also gained use in the synthesis of, e.g., antihyperglycemic, anticancer, and wound healing drugs and PDE inhibitors, which spells a potential huge importance of 4-Bromomethyl-2-cyanobiphenyl both in the development and production of new pharmaceutical compounds. This is in addition to the importance of it in new production sites for the already commercialized derivative of it with expired patents. A convenient, green method to synthesize 4-Bromomethyl-2-cyanobiphenyl was developed, where visible light is used to initiate the radical chain reaction and the H2O2/HBr system as the bromine source. A range of solvents was tested, and a final multigram protocol was developed to give a good yield of 71% of product with 97% purity and no other purification than simple filtration and wash. Diethyl carbonate was found to be an efficient greener solvent alternative to classical solvents such as carbon tetrachloride and chlorobenzene, and 2-propanol, also considered a green solvent, was suitable for precipitation and washing in the purification step.[2]
References
[1]Yang, J., Wu, H., Wang, Y., Luan, Q., Zhang, J., Wang, G., & Hao, H. (2015). Thermodynamics of 4'-bromomethyl-2-cyanobiphenyl in different solvents. The Journal of Chemical Thermodynamics, 83, 77-84.
[2]Sveegaard, S. G. (2024). A Facile Method for Multigram Scale Light-Induced Synthesis of 4'-(Bromomethyl)-2-cyanobiphenyl. Organic Process Research & Development, 28(4), 1055–1060.
You may like
Lastest Price from 4-Bromomethyl-2-cyanobiphenyl manufacturers
US $0.00/kg2025-09-24
- CAS:
- 114772-54-2
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- 1000kgs
US $0.00/kg2025-09-04
- CAS:
- 114772-54-2
- Min. Order:
- 1kg
- Purity:
- 99%min
- Supply Ability:
- 20tons