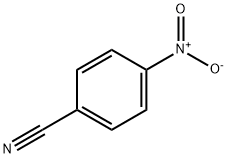
4-Nitrobenzonitrile: Versatile Aromatic Intermediate
4-Nitrobenzonitrile is a valuable and versatile chemical intermediate with significant applications in the synthesis of pharmaceuticals, agrochemicals, and other specialty chemicals. Its dual functionality allows for a wide range of chemical transformations, making it a key building block in organic synthesis. 4-Nitrobenzonitrile has been investigated for its potential as a radiosensitizer in cancer therapy. Radiosensitizers are compounds that make tumor cells more susceptible to radiation treatment. The mechanism is believed to involve the formation of radical adducts with α-alkoxyalkyl radicals generated during the radiolysis of water.

Solubility Determination and Modeling for 4-Nitrobenzonitrile in Binary Solvent
4-Nitrobenzonitrile (4NT) is an important aromatic nitrile which is generally widely used for the pharmaceuticals, dyestuffs, and pesticides production. At present, it is commonly produced by benzonitrile nitration with HNO3 or other nitrating agent. Nevertheless, during the nitration process of benzonitrile, the 3-nitrobenzonitrile isomer is also generated simultaneously as a byproduct. So further use of 4NT is limited in some fields because of the isomeric byproduct. As the rapid development of dyestuff industry and pharmaceutical industry, the required purity of 4NT is becoming very high. In general, the 4NT production is made in some solvents, for instance, acetonitrile, acetone and methanol. The 3/4-nitrobenzonitrile solubility in these solvents influences greatly the yield of product and reaction rate. The separation for organic isomers has been attracting extensive attention owing to the great requirements and importance in industry. Nevertheless, due to their similar boiling points, it is not successfully realized by using the distillation technique. It is well-known that the solid solubility in pure solvents and solvent mixtures is of significant importance during the process of solvent crystallization. As a result, the solubility is significant in the purification process of 4NT through crystallization. In order to remove 3-nitrobenzonitrile from crude 4NT product, the knowledge of 3/4-nitrobenzonitrile solubility in different neat solvents and solvent mixtures is a crucial procedure. It is known to us that mixed solvents with temperature change is a common method to change solubility of a solid. The knowledge concerning solubility in solvent mixtures can enable us to discover suitable solvents in purifying 4NT.[1]
4-Nitrobenzonitrile solubility in mixed solvents of ethyl acetate (1) + (ethanol, methanol, isopropanol, and n-propanol) (2) measured via isothermal equilibrium technique was reported covering the temperatures ranging from 278.15 to 318.15 K under p = 101.1 kPa. It increased with increasing temperature and ethyl acetate composition, and the maximum solubility value was found in ethyl acetate. The dependence of mole fraction solubility of 4-nitrobenzonitrile upon solvent composition and temperature was described through the Apelblat–Jouyban–Acree model, van’t Hoff–Jouyban–Acree model, and Jouyban–Acree model. The obtained root-mean-square deviations and mean relative deviations were, respectively, lower than 2.13% and 11.72 × 10–4 for correlative studies.
Study of 4-Nitrobenzonitrile Adsorbed on Ag and Ag/Pd Nanoparticles
Metal colloidal nanoparticles have found many applications in chemistry, mainly concerning trace detection and heterogeneous catalysis. Surface-enhanced Raman scattering (SERS) is a powerful tool for the investigation of “in situ” catalytic reactions allowing the detection of both surface-adsorbed reagents and products. In fact, for coinage metal substrates, constituted by colloidal nanoparticles or by nanostructured surfaces, huge enhancements (up to 106−107 times) of the Raman signal of the adsorbed molecules can be observed. To avoid instability problems of these colloids, we deposited the Ag/Pd particles on quartz, obtaining SERS-active nanostructured substrates, which showed sizable catalytic activity in hydrogenation reactions of nitroarenes such as 4-nitro,3-pyrazole carboxylic acid (NPC) or 4-nitrobenzonitrile (NBN). For the first compound the formation of the aminoderivative was ascertained by reduction of the nitrogroup, whereas for NBN the formation of the azoderivative was proposed on the basis of the SERS results. Here, the adsorption and the reactivity of NBN on both pure Ag and Ag/Pd bimetallic particles immobilized on quartz plates have been studied by coupling SERS measurements performed by micro-Raman apparatus and density functional theory (DFT) calculations. To interpret the SERS data, a preliminary assignment of the Raman data of NBN has been carried out. In fact no Raman data have been previously reported in the literature for this compound. The complete assignment of all the vibrational bands of 4-Nitrobenzonitrile has been performed with the help of DFT calculations.[2]
4-Nitrobenzonitrile does not show absorption in the visible spectral region but below 350 nm. This implies that no significant electronic conjugation between aromatic ring and substituents exists for this molecule, contrary to what is observed for other nitroarenes such as PNA, a typical “push−pull” compound, where a marked intramolecular charge-transfer between aminogroup (electron donor) and nitrogroup (electron acceptor) occurs. Palladium, as well as platinum and nickel, is widely employed in catalytic reactions. These metals, however, are not generally selective for the reduction of nitrogroups; for this reason, many efforts were performed for fabricating new catalytically efficient materials, mainly by mixing different metals and metal oxides. For example, gold catalysts, obtained with TiO2 or Fe2O3, recently allowed the reduction of 4-Nitrobenzonitrile to the corresponding amine with high yields and selectivity. Here, instead, we have prepared bimetallic Ag/Pd nanoparticles, which are able to reduce selectively nitrogroups to azogroups, even in the presence of other reducible functions like cyanogroup, as in the case of NBN. In addition, these nanomaterials provide satisfactory SERS enhancement to detect both reactants and reaction products. The DFT approach, employed in the present study, has provided very useful information for the assignment of the SERS spectra obtained from both pure Ag and Ag/Pd nanostructured substrates. Hence, reliable conclusions can be proposed about the species obtained by hydrogenation of 4-Nitrobenzonitrile, on the adsorption processes and on the reduction mechanism promoted by the presence of palladium in the bimetallic substrates.
References
[1]Wanmin, L., Li, M., Wang, N., Fei, Z., Chen, J., & Zhao, H. (2018). Solubility determination and modeling for 4-nitrobenzonitrile in binary solvent mixtures of ethyl acetate plus (methanol, ethanol, n-propanol, and isopropanol). Journal of Chemical & Engineering Data, 63(10), 3933–3940. https://doi.org/10.1021/acs.jced.8b00555
[2]Muniz-Miranda, M., Pergolese, B., & Bigotto, A. (2008). Surface-Enhanced Raman Scattering and Density Functional Theory Study of 4-Nitrobenzonitrile Adsorbed on Ag and Ag/Pd Nanoparticles. The Journal of Physical Chemistry C, 112 1, 6988–6992. https://doi.org/10.1021/JP711745T
You may like
See also
Lastest Price from 4-Nitrobenzonitrile manufacturers
US $0.00-0.00/KG2025-06-24
- CAS:
- 619-72-7
- Min. Order:
- 1KG
- Purity:
- 99.0%
- Supply Ability:
- 10000KGS

US $10.00/KG2025-04-21
- CAS:
- 619-72-7
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt