5-Bromouracil: Mutagenic DNA Base Analogue
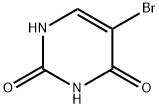
5-bromouracil is a synthetic analogue of the pyrimidine base uracil, where the hydrogen atom at the 5th position is replaced by a bromine atom. This structural modification allows it to be incorporated into DNA, leading to various mutagenic effects. 5-bromouracil has been extensively utilized in genetic engineering and mutation studies due to its well-characterized mutagenic properties. The use of 5-bromouracil as a model compound has provided valuable insights into how structural modifications to DNA bases can alter their hydrogen bonding patterns and lead to the misincorporation of nucleotides during replication. Additionally, studies on the effects of 5-bromouracil exposure have shed light on the broader consequences of DNA base analogues, including their potential to cause DNA damage, chromosomal aberrations, and cellular dysfunction. These insights have contributed to our understanding of the fundamental processes governing genetic stability and the complex interplay between DNA structure, replication, and the maintenance of genomic integrity.

Deactivation of 5-bromouracil after ultraviolet excitation
5-Bromouracil (5BU) can replace thymine (T) in DNA, causing strong mutagenic effects that have been explained by base mispairings due to 5BU’s tautomeric equilibrium. The substitution of T by 5BU is also accompanied by an increased ultraviolet sensitivity of the affected DNA, where the presence of 5BU can cause single-strand breaks, alkali-labile bonds, double-strand breaks and DNA–protein cross-linking. The occurrence of these lesions originates from the ability of 5-bromouracil to cleave the C–Br bond, leading to the formation of a uracilyl radical. This radical in turn can abstract a hydrogen atom from a suitable donor (e.g. an adjacent sugar), thereby forming uracil (U). Hence, the uracilyl radical is responsible for DNA damage and can be exploited for a variety of processes, such as DNA cross-linking, adenine elimination or generation of reactive oxygen species for photodynamical therapy. Mechanistically, static quantum chemical calculations on isolated 5-bromouracil have provided support for two pathways operating after 5BU is irradiated by UV light—one pathway is responsible for bromine elimination and the second leads back to the reactant through a conical intersection. According to that study, there are two internal coordinates that are critical for the description of the deactivation processes of 5-bromouracil.[1]
We performed accurate vertical excitation calculations and potential energy surface explorations for 5-bromouracil, in order to investigate the excited-state dynamics of this molecule. The vertical excitation calculations showed that MS-CASPT2 with an active space of 20 electrons in 14 orbitals gives excitation energies in excellent agreement with experiment. In particular, the lowest excited states are the bright 1ππ* state, a dark 1nOπ* state and a dissociative 1πσ* state at slightly higher energies. These three states are responsible for the three plausible photophysical reaction pathways of 5-bromouracil, according to MS-CASPT2. A barrierless pathway on the 1ππ* state leads directly to a conical intersection with the S0, allowing for efficient relaxation to the ground state. We also investigated the performance of different electronic structure methods in describing the deactivation of 5-bromouracil, including ADC(2), TD-BP886, TD-B3LYP, SA-CASSCF and MRCIS. All of these methods represent significantly lower cost alternatives to the expensive MS-CASPT2 computations performed. From the excitation energies and potential energy surfaces, ADC.
We found that ISC is mediated by the 1nOπ* state and occurs with a time constant of 450±100 fs, showing that this process is ultrafast in 5-bromouracil, as already discussed in recent literature on pyrimidine bases. Unfortunately, the trajectories do not allow one to obtain yields for the competitive photophysical pathways of 5BU, due to the shortcomings of MRCIS. This study clearly shows that the proper description of the excited-state dynamics of 5-bromouracil is very challenging, and requires a multi-configurational electronic structure method with a large active space, in order to account for all the different, competing relaxation pathways. Moreover, previous experiments were conducted in the condensed phase, and the applications of 5-bromouracil involve the photoactive molecule in a complex biological environment, and the inclusion of such environments would add another level of complexity to future computations.
5-Bromouracil-gracillin (5BrU-G) complex
Recent cancer treatment development has focused on smart drugs, primarily using nanomaterials as carriers. However, concerns about nanomaterial fate and body clearance have led to exploring alternative approaches. This study presents a novel targeted smart drug that uses normal lymphocytic cells as carriers and exploits cancer microenvironment characteristics for drug release, avoiding systemic damage. The research investigated a complex combining gracillin (natural carrier) and the chemotherapeutic agent 5-bromouracil (5-BrU). Molecular docking showed the 5-Bromouracil-gracillin complex had superior binding affinity (- 7.96 kcal mol-1) to glycosylated adhesion domain of human T lymphocyte glycoprotein CD2 (1CDB) cell surface receptors in silico. The complex was successfully synthesized through double replacement, precipitation, and neutralization reactions, confirmed by liquid chromatography-tandem mass spectrometry (LC-MS/MS).[2]
Cytotoxic and genotoxic studies revealed the selectivity of 5-Bromouracil-gracillin against cancer cells (MDA-MB-231 and Caco-2) while saving normal cells (MCF-10A and CCD 841 CoN). Unlike 5-Bromouracil alone, which showed significant genotoxicity in normal cells, the 5BrU-G complex demonstrated minimal toxic effects. The selective targeting mechanism of 5-Bromouracil-gracillin relies on APOBEC3 enzyme activity, which is elevated in cancer cells but is absent in normal cells. This was confirmed when APOBEC3 inhibition prevented the complex's cancer-killing activity. This novel approach offers promising alternatives for improving cancer therapy efficacy while reducing side effects.
References
[1]Peccati F, Mai S, González L. Insights into the deactivation of 5-bromouracil after ultraviolet excitation. Philos Trans A Math Phys Eng Sci. 2017 Apr 28;375(2092):20160202. doi: 10.1098/rsta.2016.0202. PMID: 28320905; PMCID: PMC5360901.
[2]Hatim MS, Al-Saffar AZ, Al-Aadhami MAWS. 5-Bromouracil-gracillin (5BrU-G) complex: an APOBEC3-activated therapeutic strategy exploiting cancer-specific enzymatic activity for selective cytotoxicity. Med Oncol. 2025 May 8;42(6):203. doi: 10.1007/s12032-025-02745-x. PMID: 40335833.
You may like
See also
Lastest Price from 5-Bromouracil manufacturers

US $0.00/kg2025-09-01
- CAS:
- 51-20-7
- Min. Order:
- 1kg
- Purity:
- 99%min
- Supply Ability:
- 20tons

US $0.00/KG2025-04-21
- CAS:
- 51-20-7
- Min. Order:
- 1KG
- Purity:
- ≥98.0%
- Supply Ability:
- 100kg/month