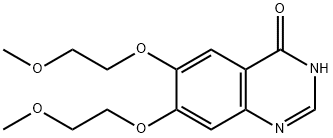
6,7-Bis-(2-methoxyethoxy)-4(3H)-quinazolinone:synthesis and new application
Introduction
Quinazolinones have been widely used in medicine and agriculture. In medicine, quinazolinones are used as anti-bacterial,anti-tumor, anti-HIV1, anti-pyretic, anti-inflammatory, anticonvulsant, and anti-parkinson’s syndrome drugs. In the field of agriculture, quinazolinones show multifarious activities including insecticidal and bactericidal activities. However,there are few reports on the tyrosinase inhibition and antibrowning effect of quinazolinones. 6,7-Bis-(2-methoxyethoxy)-4(3H)-quinazolinone is a key intermediate for the synthesis of quinazoline-based antitumor drugs such as erlotinib hydrochloride.The inhibitory mechanism of 6,7-bis-(2-methoxyethoxy)-4(3H)-quinazolinone (BMEQ) (Fig. 1)on tyrosinase was investigated. To evaluate its potential application in the food industry, the anti-browning effect of BMEQ on fresh-cut apples was further investigated.[1]

Synthesis of 6,7-Bis-(2-methoxyethoxy)-4(3H)-quinazolinone
Report 1:In order to prepare the key quinazoline intermediate 6,7-Bis-(2-methoxyethoxy)-4(3H)-quinazolinone with better antitumor activity, the method of etherification,nitration, reduction and tcyclization was used for synthesizing 6,7-Bis-(2-methoxyethoxy)-4(3H)-quinazolinone by the four steps from ethyl 3,4-dihydrxybenzoate as the starting material. The total yield was more than 90% through the improved and optimized process. The improved process is a high yield, simple operation, environment friendly and suitable for scale production.[2]
Report 2: 6,7-Bis-(2-methoxyethoxy)-4(3H)-quinazolinone was prepared through etherification, nitration,reduction and cyclization from the starting material ethyl 3,4-dihydroxybenzoate.The synthetic process was improved, and the optimum conditions for the reaction were determined with an overall yield of 83.7%.[3]
Inhibitory effect of 6,7-bis-(2-methoxyethoxy)-4(3H)-quinazolinone on tyrosinase activity
The inhibitory effect of 6,7-bis-(2-methoxyethoxy)-4(3H)-quinazolinone on tyrosinase was first investigated using enzyme assay. The result revealed that the BMEQ inhibited tyrosinase in a dose-reliant manner. Compared with cefazolin (IC50=7.0 mM) , norfloxacin (IC50=0.7 mM), and moxifloxacin (IC50=1.2 mM), the IC50 of 6,7-bis-(2-methoxyethoxy)-4(3H)-quinazolinone was 160±6 μM, which showed that 6,7-bis-(2-methoxyethoxy)-4(3H)-quinazolinone was a good tyrosinase inhibitor.
Inhibition mechanism of BMEQ on tyrosinase
In each experimental group of enzyme assay, the initial concentration of substrate (3,4-dihydroxyphenylalanine) was kept unchanged, while the initial concentration of tyrosinase was changed, so as to determine the inhibitory mechanism of 6,7-bis-(2-methoxyethoxy)-4(3H)-quinazolinone on the enzyme. The result showed that all the lines intersected at the origin, implying BMEQ was a reversible inhibitor of tyrosinase. The presence of 6,7-bis-(2-methoxyethoxy)-4(3H)-quinazolinone did not result in a decrease in enzyme quantity, but caused a decline in enzyme activity.[1]
Inhibition type of BMEQ on tyrosinase
The inhibitory type of 6,7-bis-(2-methoxyethoxy)-4(3H)-quinazolinone on tyrosinase was determined by changing the concentration of the substrate, while keeping that of the enzyme constant.The result showed that a group of straight lines with different slopes intersected at a point on the Y axis, indicating that 6,7-bis-(2-methoxyethoxy)-4(3H)-quinazolinone is a competitive inhibitor of tyrosinase. Based on the findings, a linear relationship between the slopes and the inhibitor concentrations was observed. This indicated that tyrosinase possessed either one binding site or a specific type of binding site for the 6,7-bis-(2-methoxyethoxy)-4(3H)-quinazolinone. Moreover, the inhibitory constant (KI) was evaluated to be 99±3 μM. In comparison to naringin (KI=3.04 mM), salicylic acid (KI=2.49 mM), and 2-hydroxycinnamic acid (KI=0.45± 0.05 mM), BMEQ exhibited a lower KI value, which indicated that BMEQ have a higher binding affinity towards the free enzyme and thereby inhibiting tyrosinase activity dramatically.[1]
Effect of BMEQ on the contents of total phenols and flavonoids in fresh-cut apples
The study indicated the content of total phenols in fresh-cut apples with no special treatment decreased rapidly during the total storage time.Compared with the control group, the group that treated with 6,7-bis-(2-methoxyethoxy)-4(3H)-quinazolinone maintained at a higher content of total phenols. On day 8,the total phenolics content in apple slices treated with 0.25, 0.5,1, and 3 mM 6,7-bis-(2-methoxyethoxy)-4(3H)-quinazolinone was 1.5, 1.6, 1.9, and 2.0 times higher than the control group, respectively. The result indicated that the BMEQ delayed the oxidation of phenols in fresh cut apples. Research showed that the flavonoid content of fresh cut apples decreased rapidly during the whole storage time. Under the treatment of 6,7-bis-(2-methoxyethoxy)-4(3H)-quinazolinone, a significantly higher content of flavonoid was observed compared with the control group, with a positive correlation between the concentrations of 6,7-bis-(2-methoxyethoxy)-4(3H)-quinazolinone and the content of flavonoids in fresh-cut apple. The result showed that BMEQ could markedly inhibit the polymerization of apple flavonoids. In summary, BMEQ delayed the oxidation of total phenols and flavonoids, and thus controlled the browning of fresh-cut apples.
In addition, the 6,7-bis-(2-methoxyethoxy)-4(3H)-quinazolinone postponed the oxidation of phenolics and flavonoids by inhibiting polyphenol oxidase (PPO) and peroxidase (POD),which resulted in the inhibition of the browning of fresh-cut apples. This study identified a novel tyrosinase inhibitor BMEQ and verified its potential application for improving the preservation of postharvest fruits.
References
[1] Chai WM, Bai Q, Pan Q, Wang L, Zhu D. 6,7-Bis-(2-methoxyethoxy)-4(3H)-quinazolinone as a novel inhibitor of tyrosinase and potential anti-browning agent of fresh-cut apples. J Biosci Bioeng. 2024;137(3):165-172. doi:10.1016/j.jbiosc.2023.12.006
[2] Sun HL,et al., Study on the synthesis of 6,7-Bis-(2-methoxyethoxy)-4(3H)-quinazolinone[J].Journal of Natural Science of Heilongjiang University,2011,28(02):223-228.DOI:10.13482/j.issn1001-7011.2011.02.006.
[3] Chen HQ,et al., Synthesis of 6,7-Bis-(2-methoxyethoxy)-4(3H)-quinazolinone[J].Chemical World,2010,51(04):224-227+231.DOI:10.19500/j.cnki.0367-6358.2010.04.010.
You may like
See also
Lastest Price from 6,7-Bis-(2-methoxyethoxy)-4(3H)-quinazolinone manufacturers

US $0.00-0.00/kg2024-11-26
- CAS:
- 179688-29-0
- Min. Order:
- 1kg
- Purity:
- 99%(HPLC)
- Supply Ability:
- 2000kg

US $10.00-7.00/kg2024-08-20
- CAS:
- 179688-29-0
- Min. Order:
- 1kg
- Purity:
- 99.9%
- Supply Ability:
- 20ton