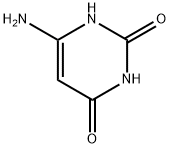
6-Aminouracil: Heterocycle Synthesis & Nucleodye Development
6-Aminouracil is a significant pyrimidine derivative. ts molecule exhibits both nucleophilic and electrophilic reactivity, making it a core intermediate in pharmaceutical and fine chemical industries. It is extensively employed in synthesising bioactive compounds containing heterocycles. As a versatile pyrimidine analog, 6-aminouracil serves as a crucial scaffold in medicinal chemistry for the development of novel therapeutic agents, particularly in the realm of oncology and infectious diseases.

5-Amino- and 6-amino-uracils in the synthesis of various heterocycles
Uracil is a naturally occurring pyrimidine-based compound, which is considered one among the four nucleobases in RNA. The chemical characteristics of uracil and its derivatives, such as 5-aminouracils and 6-aminouracils, vary because these molecules have the ability to function as both electrophiles and nucleophiles. Numerous bioactive chemical compounds can be synthesized using 5-aminouracils and/or 6-aminouracils as precursors. Because of their synthetic accessibility and a variety of biological actions, uracil derivatives are regarded as preferred structures in drug discovery. Particularly, 5- and 6-aminouracils and their derivatives have revealed various biological properties such as antioxidant, antimicrobial, anticancer, anti-Alzheimer and antiviral activity. Pyrido-, pyrrolo- and pyrimido-pyrimidines, phenazine, chromenes and xanthines of 5- and 6-aminouracils and their derivatives have been reported to be biologically active compounds. In this review, we discuss the synthesis of heterocycles using 5-amino- and 6-amino-uracils over the past 10 years, especially from 2014 to 2024. We also provide more insight into the multi-component reaction in one-pot process pertaining to the synthesis of target compounds. Some reaction mechanisms are also covered. We also address the biological significance of target heterocycles as therapeutic agents. [1]
Due to the biological activity of 5-amino uracil, 6-aminouracil and their derived heterocycles, their synthesis has attracted a great interest in the field of medicinal chemistry. This study includes different methods reported for synthesizing the target molecules in the past 10 years, especially from 2014 to 2024. We also reported on the advantages of catalysis in synthesizing the target compounds using a multi-component reaction (MCR) sequence. Accordingly, we here shed light on the utility of catalysis in MCRs, which would provide the target molecules with excellent yields in addition to short reaction time, cost efficiency, and a simple workup procedure without extra purification techniques. This review also focused on the biological applications of the target molecules since they covered many biological activities, such as anti-cancer, anti-microbial, anti-inflammatory, anti-Alzheimer, and/or anti-tubercular agents. Due to the therapeutic importance of heterocycles from 5-amino and 6-aminouracils, their synthesis has become valuable in the synthesis of numerous drugs. The synthesis of the former compounds has become more facile due to the utility of various eco-friendly catalysts in multicomponent reactions. Moreover, mild reaction conditions, good to excellent yields, and the absence of tedious separation procedures are the key advantages of their synthesis. The latter would encourage researchers to synthesize former heterocycles, which would lead to interesting biological applications.
Synthesis and Characterization of 6-Aminouracil-Based Nucleodyes
The ongoing challenge of studying nucleic acid and their cellular interactions, while minimally perturbing their constituents, has led to a growing interest in fluorescent nucleoside probes. The development of such emissive analogues has been triggered by the nonemissive nature of the natural nucleobases and has been accelerated by the increasing availability of highly sensitive fluorescence-based techniques. Current efforts have concentrated on the development of smaller and responsive probes, displaying higher isomorphicity and sensitivity to polarity as well as favorable red-shifted absorption and emission bands. In the current study, we exploit the versatility of 6-aminouracil to obtain both uridine and cytidine azo dye analogues. We prepared both 6-amino-5-(4-nitrophenylazo)-uridine (5) and 5-(4-nitrophenylazo)-6-oxocytidine as new pyrimidine-based nucleodyes derived from 6-aminouracil. Their structural and photophysical features are characterized as well as their responsiveness to microenvironmental changes including polarity and pH. Our observations are supplemented by quantum chemical calculations of absorptive properties, which nicely predict these compounds to be highly absorbing visible dyes. We demonstrate that small nucleoside-based quenchers can be designed to electronically match emissive nucleosides and other known fluorescent probes.[2]
The assembly of a uracil azo dye is influenced by the nature of the substituent on the phenylazo chromophore. Electron-donating phenylazo substitution at the 6 position could conceivably be constructed by diazotization of 6-aminouracil and subsequent reaction with electron-rich substituted benzene. Electron-withdrawing phenylazo substitution at the 5 position could be constructed differently by nucleophilic attack of the 5 position on a substituted benzenediazonium ion. Indeed, uracil reacts under basic conditions with benzenediazonium ion to give 5-phenylazouracil derivatives. However, 6-aminouracil does not react under diazotation conditions to give the corresponding uracil-6-diazonium salt, but rather yields the nitrosylation product at the highly nucleophilic 5 position. 6-Aminouracil also readily reacts with benzenediazonium ions to afford 6-amino-5-phenylazouracil derivatives. Starting from 6-aminouracil, two pyrimidine nucleodyes were prepared and studied for their potential as visible-range probes. Crystal structure analysis indicates that both 6-amino-5-(4-nitrophenylazo)uridine and 5-(4-nitrophenyl-hydrazono)-6-oxocytidine are good structural analogues of their native nucleoside counterparts. We recognize that the use of these nucleodyes as visible-range probes might be limited as the impact of pH and polarity on their photophysical features indicates that microenvironmental changes might be difficult to distinguish.
References
[1]Aly AA, Osman EM, Mostafa SM, Bedair TM, Abd-Elmonem M, Sadek KU, Mohamed AH. 5-Amino- and 6-amino-uracils in the synthesis of various heterocycles as therapeutic agents: a review. RSC Adv. 2025 Oct 20;15(46):39235-39287. doi: 10.1039/d5ra05123a. PMID: 41122147; PMCID: PMC12536882.
[2]Freeman NS, Moore CE, Wilhelmsson LM, Tor Y. Chromophoric Nucleoside Analogues: Synthesis and Characterization of 6-Aminouracil-Based Nucleodyes. J Org Chem. 2016 Jun 3;81(11):4530-9. doi: 10.1021/acs.joc.6b00310. Epub 2016 May 17. PMID: 27128151; PMCID: PMC5493935.
You may like
Lastest Price from 6-Aminouracil manufacturers

US $25.00/ASSAYS2025-04-21
- CAS:
- 873-83-6
- Min. Order:
- 100ASSAYS
- Purity:
- 99.5%
- Supply Ability:
- 100 mt

US $0.00-0.00/kg2025-04-04
- CAS:
- 873-83-6
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- 1Ton