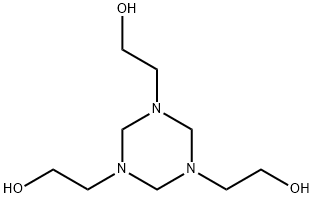
Analysis and application research of hexahydro-1,3,5-tris(hydroxyethyl)-s-triazine
Introduction
Hexahydro-1,3,5-tris(hydroxyethyl)-s-triazine(Figure 1), also known as hexahydro-1,3,5-tris(2-hydroxyethyl)-s-triazine, is a widely used biocide that is added to water-based metalworking fluids (MWF) to control populations of microorganisms. Uncontrolled concentrations of this biocide in MWF have been implicated in recent outbreaks of hypersensitivity pneumonitis, possibly because it may induce development of biocide-resistant mycobacteria and subsequent overgrowth of these organisms in MWF sumps even as it eliminates other, competitive microorganism populations. The mycobacteria are suspected agents in the induction of pneumonitis.[1]

Analysis of hexahydro-1,3,5-tris(hydroxyethyl)-s-triazine in metalworking fluids
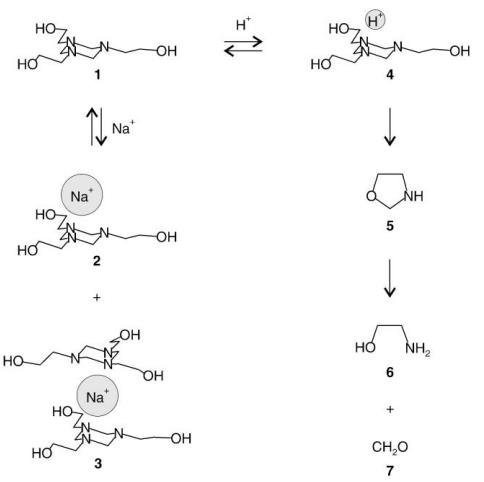
As shown in Fig. 2, hexahydro-1,3,5-tris(hydroxyethyl)-s-triazine(1) is readily protonated under acidic conditions (which may exist in the immediate vicinity of target microorganisms due to metabolic activity) to form a relatively unstable species (4), which is believed to decompose through an oxazolidine intermediate (5) to ethanolamine (6)and formaldehyde (7). The latter is presumably the actual antimicrobial agent. To provide optimal treatment of MWF sumps with hexahydro-1,3,5-tris(hydroxyethyl)-s-triazine, an analytical method is needed to assess the residual concentration of the compound in MWF. However,there appear to be no methods available for direct measurement of hexahydro-1,3,5-tris(hydroxyethyl)-s-triazine. In-house attempts to analyze TA via gas chromatography have been unsuccessful due to its thermal lability. In a recent study of MWF, the analyte was measured indirectly by conversion to formaldehyde, which was derivatized to form diacetyl dihydrolutidine and analyzed by high performance liquid chromatography (HPLC) with diode array detection.[1]
Hexahydro-1,3,5-tris(hydroxyethyl)-s-triazine is a widely used biocide in metalworking fluids that resists direct quantification in many analytical methods due to instability. It can be detected in electrospray-mass spectrometry (ES-MS) due to the formation of a charged and relatively stable adduct with the sodium ion. This adduct produces a distinct ion spectrum via collision-induced fragmentation, which should promote specific detection of the analyte in complex matrices. ES-MS detection of the analyte added to, or already present in,metalworking fluid samples at ug/mL levels is demonstrated. Parameters affecting the formation and detection of the sodium adduct, including choice of solvent, alkalinity, and sodium ion level are explored.Linearity of response in flow injection mode is demonstrated.[1]
Application of hexahydro-1,3,5-tris(hydroxyethyl)-s-triazine
For H2S Scavenging
Natural gas produced worldwide contains, among other compounds, H2S and CO2. Both gases in the presence of free water can generate problems of corrosion in pipes and subtract calorific value to this gaseous hydrocarbon. Natural gas containing H2S and CO2 above the permissible limits should be treated to remove these components. Particularly, H2S is removed mainly using hexahydro-1,3,5-tris(hydroxyethyl)-s-triazine (TZ), which is the liquid scavenger of H2S leading on the market. By means of computational methods, researchers have studied the reaction of H2S with hexahydro-1,3,5-tris(hydroxyethyl)-s-triazine, oxazolidine, and oxazinane to elucidate, explain, or confirm different aspects related to their proposed reaction mechanisms. These results validate thermodynamically the production of hexahydro-1,3,5-tris(hydroxyethyl)-s-triazine from MEA and formaldehyde, justify the presence of mixtures of TZ and oxazolidine in some commercial products, and explain why in the complexity of the H2S scavenging mechanism with hexahydro-1,3,5-tris(hydroxyethyl)-s-triazine, tritiane is not formed and dithiazine will prefer to react by polymerization to form amorphous dithiazine and that the scavenging capacity for H2S should follow the TZ ≫ oxazinane > oxazolidine sequence.[2]
Differential biocide susceptibility of the multiple genotypes of Mycobacterium immunogenum
The non-tuberculous mycobacterium Mycobacterium immunogenum colonizes industrial metalworking fluids (MWFs) presumably due to its relative resistance to the currently practiced biocides and has been implicated in occupational respiratory hazards, particularly hypersensitivity pneumonitis. With an aim to understand its inherent biocide susceptibility profile and survival potential in MWF, five different genotypes of this organism, including a reference genotype (700506) and four novel test genotypes (MJY-3, MJY-4, MJY-10 and MJY-12) isolated in our recent study from diverse MWF operations were evaluated. For this, two commercial biocide formulations, Grotan (Hexahydro-1,3,5-tris(hydroxyethyl)-s-triazine) and Kathon (5-chloro-2-methyl-4-isothiazolin-3-one) currently practiced for the control of microorganisms, including mycobacteria, in MWF operations were tested. Effect of the fluid matrix on the biocide susceptibility was investigated for the synthetic (S) and semi-synthetic (SS) MWF matrices. In general, the minimum inhibitory concentration values were higher for the HCHO-releasing biocide Grotan than the isothiazolone biocide Kathon. All genotypes (except the reference genotype) showed lower susceptibility in SS as compared to S fluid matrix for Grotan(Hexahydro-1,3,5-tris(hydroxyethyl)-s-triazine). However, in case of Kathon, a greater susceptibility was observed in SS fluid for majority of the test genotypes (MJY-3, 4 and 10). The test genotypes were more resistant than the reference genotype to either biocide in both fluid types. Furthermore, the individual genotypes showed differential biocidal susceptibility, with MJY-10 being the most resistant. These observations emphasize the importance of using the resistant genotypes of M. immunogenum as the test strains for formulation or development and evaluation of existing and novel biocides, for industrial applications.[3]
References
[1] Pretty J, Glaser R, Jones J 3rd, Lunsford RA. A technique for the identification and direct analysis of hexahydro-1,3,5-tris(2-hydroxyethyl)-s-triazine in metalworking fluids using electrospray-mass spectrometry. Analyst. 2004;129(11):1150-1155. doi:10.1039/b404578e
[2] Román MN, Díaz MA, Coll DS. Study of the Reaction Mechanism of Triazines and Associated Species for H2S Scavenging. ACS Omega. 2023;8(13):12165-12172. Published 2023 Mar 21. doi:10.1021/acsomega.2c08103
[3] Selvaraju SB, Khan IU, Yadav JS. Differential biocide susceptibility of the multiple genotypes of Mycobacterium immunogenum. J Ind Microbiol Biotechnol. 2008;35(3):197-203. doi:10.1007/s10295-007-0286-5
You may like
Lastest Price from Hexahydro-1,3,5-tris(hydroxyethyl)-s-triazine manufacturers
US $0.00-0.00/KG2025-12-05
- CAS:
- 4719-04-4
- Min. Order:
- 1KG
- Purity:
- 98
- Supply Ability:
- 10000KGS

US $6.00/kg2025-04-21
- CAS:
- 4719-04-4
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 2000KG/Month