Application examples of 6-bromohexanoic acid
Introduction
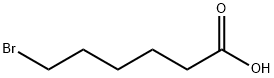
6-Bromohexanoic acid (Figure 1) is a halogenated aliphatic carboxylic acid with the molecular formula C₆H₁₁BrO₂. It serves as a versatile intermediate in organic synthesis, widely used for the preparation of functionalized compounds, polymers, and pharmaceutical intermediates. 6-Bromohexanoic acid features a bromine atom at the terminal carbon of a six carbon alkyl chain, making it valuable for nucleophilic substitution reactions and the introduction of carboxyl-containing alkyl groups into target molecules.

A promising hybrid nano-gene-delivery vector prepared
Polyethylenimine (PEI) is one of the most widely used polymers in gene delivery. The aim of this study was to modify PEI by replacing some of its primary amines with Brevinin 2R (BR-2R) peptide in order to increase the efficiency of gene delivery. Polyethylenimine was modified by BR-2R peptide by two different approaches; A) conjugation methods including (І) using succinimidyl 3-(2-pyridyldithio) propionate (SPDP), (П) EDC/NHS protocol and (ПІ) EDC/NHS+6-bromohexanoic acid protocol, and B) physical interaction method. The modified polymers were characterized for their ability of plasmid condensation, number of primary amines, size and zeta potential. The transfection efficiency and cytotoxicity were evaluated on HEK293, L929, WEHI164 and Neuro2A cell lines by green fluorescent protein (GFP)-based plasmid (pGFP) reporter gene and viability assays, respectively. Apoptosis induction ability was also evaluated via PI/Annexin V assay. Polyplex had size and zeta potential between 200-270 nm and +21.5- +28.4 mV, respectively. All vectors were able to condense plasmid DNA in C/P=4 (carrier-plasmid ratio). Transfection results on the Neuro2A cell line showed that the vector containing the BR-2R peptide, which was synthesized using EDC-NHS protocol had the best transfection efficiency. These results showed that conjugation of Brevinin 2R as cell penetrating peptide to polyethyleneimine could enhance the transfection ability of the polymer.[1]
Surface Functionalization of Metals by Alkyl Chains
Alkyl chains are covalently attached onto metal surfaces by indirect reduction of the bromoalkyl derivative (RBr). This indirect reaction involves the formation (by spontaneous or electrochemical reduction of the 2,6-dimethylbenzenediazonium salt) of a sterically hindered aryl radical that abstracts a Br atom from RBr but does not react with the surface. This crossover reaction furnishes an alkyl radical that reacts with the surface. Starting from 6-bromohexanoic acid, carboxylic functionalized gold surfaces are prepared. "Layer-by-layer" assemblies are built from these surfaces and present some ionic selectivity.[2]
6-bromohexanoic acid (6BH) can induce an immune response
Lipoylated enzymes such as the E2 component of pyruvate dehydrogenase complex (PDC-E2) are targets for autoreactive immune responses in primary biliary cirrhosis, with lipoic acid itself forming a component of the dominant auto-epitopes. A candidate mechanism for the initiation of tolerance breakdown in this disease is immune recognition of neo-antigens formed by xenobiotic substitution of normal proteins. Importantly, sensitization with proteins artificially substituted with the lipoic acid analogue xenobiotic 6-bromohexanoic acid (6BH) can induce an immune response that cross-reacts with PDC-E2. This study investigated the potential of recombinant lipoylation enzymes lipoate activating enzyme and lipoyl-AMP(GMP):N-lysine lipoyl transferase to aberrantly incorporate xenobiotics into PDC-E2. It was found that these enzymes could incorporate lipoic acid analogues including octanoic and hexanoic acids and the xenobiotic 6-bromohexanoic acid into PDC-E2. The efficiency of incorporation of these analogues showed a variable dependence on activation by adenosine triphosphate (ATP) or guanosine triphosphate (GTP), with ATP favoring the incorporation of hexanoic acid and 6BH whereas GTP enhanced substitution by octanoic acid. Importantly, competition studies showed that the relative incorporation of both 6-bromohexanoic acid and lipoic acid could be regulated by the balance between ATP and GTP, with the formation of 6-bromohexanoic acid-substituted PDC-E2 predominating in an ATP-rich environment.
Using a well-defined system in vitro we have shown that an important xenobiotic can be incorporated into PDC in place of lipoic acid by the exogenous lipoylation system; the relative levels of lipoic acid and xenobiotic incorporation may be determined by the balance between ATP and GTP. These observations suggest a clear mechanism for the generation of an auto-immunogenic neo-antigen of relevance for the pathogenesis of primary biliary cirrhosis.[3]
Modified polyethyleneimine with histidine-lysine short peptides
There are several strategies that can be utilized to improve transfection efficiency while reducing the cytotoxicity of polyethyleneimine (PEI) as a promising non-viral gene delivery vector. In this study, we evaluated the potential use of lysine-histidine (KH) peptides in modifying the PEI 10 kDa structure and enhancing its efficiency while maintaining low toxicity of PEI. PEI 10 kDa was modified with 6-bromohexanoic acid (alkyl) to increase its lipophilicity. Then, ethylenediamine (EDA) was attached to the carboxylic groups of PEI-hexanoate to restore the primary amines of PEI. Subsequently, six different KH short peptides were conjugated to PEIs and evaluated for the effect of the KH sequence on vector transfection efficiency and cytotoxicity. The transfection efficiency of PEI-peptides complexed with a luciferase reporter gene (pRLCMV) in Neuro-2A murine neuroblastoma cells showed that the PEI conjugated to KHHHKKHHHK peptide had a significantly higher rate of gene transfection efficiency in comparison with other KH peptides. This peptide was conjugated to PEI-alkyl and PEI-alkyl-EDA and significant improvement in efficiency with minimal cytotoxicity was observed. The results obtained suggest that the sequence and content of KH peptides will have a significant impact on the transfection efficiency of modified PEI 10 kDa.[4]
References
[1] Zohrab F, Asoodeh A, Jalili A, Darroudi M, Kazemi Oskuee R. Brevinin-2R-linked polyethylenimine as a promising hybrid nano-gene-delivery vector. Iran J Basic Med Sci. 2019;22(9):1026-1035. doi:10.22038/ijbms.2019.37125.8842
[2] Hetemi D, Médard J, Decorse P, et al. Surface Functionalization of Metals by Alkyl Chains through a Radical Crossover Reaction. Langmuir. 2016;32(25):6335-6342. doi:10.1021/acs.langmuir.6b01557
[3] Walden HR, Kirby JA, Yeaman SJ, Gray J, Jones DE, Palmer JM. Xenobiotic incorporation into pyruvate dehydrogenase complex can occur via the exogenous lipoylation pathway. Hepatology. 2008;48(6):1874-1884. doi:10.1002/hep.22540
[4] Hashemi M, Parhiz BH, Hatefi A, Ramezani M. Modified polyethyleneimine with histidine-lysine short peptides as gene carrier. Cancer Gene Ther. 2011;18(1):12-19. doi:10.1038/cgt.2010.57
You may like
Related articles And Qustion
See also
Lastest Price from 6-Bromohexanoic acid manufacturers

US $1.00/KG2025-04-21
- CAS:
- 4224-70-8
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 1000kg/month

US $0.00/KG2025-04-15
- CAS:
- 4224-70-8
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 500000kg