Application of N-Methylcyclohexylamine and its derivatives
Introduction
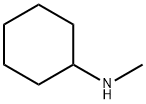
N-Methylcyclohexylamine is an organic compound belonging to the class of cycloalkylamines. It has the molecular formula C7H15N and a molecular weight of 113.20 g/mol. Typically, it appears as a colorless to pale yellow transparent liquid with a characteristic amine-like odor.(Figure 1) N-Methylcyclohexylamine is an important organic chemical raw material, which is widely used in the industrial production of pesticides, dyes, rubber additives and other products.
Synthesis of high phase transition temperature compounds based on N-methylcyclohexylamine ligands
Phase change materials have various and superior physical properties, such as ferroelectricity, switchable dielectric properties, optical effects and semiconductor properties,which show great application potential in data storage and signal processing. Therefore, the exploration of phase change materials with novel multifunctional properties and high phase change temperature has gradually become a research hotspot. In order to prepare multifunctional materials with high temperature phase transformation properties, Li et al.successfully synthesized nine novel compounds using N-Methylcyclohexylamine (MCHA) as a matrix by regulating the molecular design strategy of organic cations and inorganic metal ions. The phase transition characteristics, dielectric properties and semiconductor properties of these compounds were investigated and analyzed.
In order to find a new method to regulate the band gap, N-methylcyclohexylamine(MCHA) was used as an organic ligand to react with ZnCl2 to obtain compound(MCHA)2ZnCl4 (1) and with SbCl3 to obtain compound (MCHA)3Sb2Cl9 (2). The reaction with BiCl3 gives compound (MCHA)3Bi2Cl9 (3).(ECHA)2ZnCl4 (4) was obtained by extending the carbon chain on the basis of compound (MCHA)2ZnCl4 (1) without changing the inorganic metal ions. (MCHA)2ZnCl4 (1)was substituted by Cl/Br to obtain compound (MCHA)2ZnBr4 (5). (ECHA)2ZnCl4 (4) wassubstituted by Cl/Br to obtain compound (ECHA)2ZnBr4 (6).Based on the organic ligand (MCHA)+ in compound 1, a new organic ligand(DMCHA)+ was discovered by changing the number of carbon chains on the N atom in theorganic cation. The compounds (DMCHA)2ZnF4 (7), (DMCHA)2ZnCl4 (8) and(MCHA)2ZnBr4 (9) were obtained by reacting with ZnF2, ZnCl2 and ZnBr2, respectively. Through the study of compound structure, phase transition properties and semiconductor properties, it is found that the replacement of metalions in inorganic components changes the inorganic metal skeleton, the change of distance between H and Cl changes the interatomic force, and also changes the compound phase transition temperature and semiconductor band gap.[1]
N-Methylcyclohexylamine derivatives application
A series of U50,488 related isothiocyanates was synthesized from enantiomerically pure (S,S)-(+)-trans-2-pyrrolidinyl-N-methylcyclohexylamine [(+)-7] and (R,R)-(-)-trans-2-pyrrolidinyl-N-methylcyclohexylamine [(-)-7]. DCC coupling of (+)- and (-)-7 with nitrophenylacetic acids followed by catalytic hydrogenation and treatment with thiophosgene afforded a series of six isomeric aryl isothiocyanate analogues of U50,488. Similarly, DCC coupling of (+)- and (-)-7 with (+)- and (-)-N-t-Boc-protected phenylglycines afforded four isomeric alkyl isothiocyanates. Evaluation of the isothiocyanates for their capacity to produce wash-resistant inhibition of mu, delta, and kappa sites in vitro was performed using rat and guinea pig brain membranes. None of the compounds was able to irreversibly inhibit binding of [3H]bremazocine to guinea pig and rat brain membranes (depleted of functional mu and delta receptors by pretreatment with acylating agents BIT and FIT). However, (1S,2S)-trans-2-isothiocyanato-N-methyl-N-[2- (1-pyrrolidinyl)cyclohexyl]benzeneacetamide [(-)-1] was able to specifically and irreversibly inhibit kappa receptors labeled by [3H]-U69,593: Incubation of rat brain membranes for 60 min at 25 degrees C with 1 microM of (-)-1 resulted in a wash-resistant reduction of the binding to 11.2 +/- 2.5% of the control. Binding analysis revealed the wash-resistant reduction in [3H]-U69,593 binding by (-)-1 to be through an increase in the Kd without effect on the Bmax. (-)-1 failed to effect mu or delta binding in rat or guinea pig brain under the same conditions. The enantiomer of (-)-1, (1R,2R)-trans-2-isothiocyanato-N-methyl-N-[2- (1-pyrrolidinyl)cyclohexyl]benzeneacetamide [(+)-1], failed to affect kappa receptors labeled by [3H]-U69,593 under the same conditions as for (-)-1. (1S,2S)-trans-3-Isothiocyanato-N-methyl-N-[2- (1-pyrrolidinyl)cyclohexyl]benzeneacetamide [(-)-2] inhibited to 49.6 +/- 5.1% of the control, in a wash-resistant manner, kappa receptors labeled by [3H]-U69,593. However, (-)-2 was not as selective as (-)-1 since it also reduced [3H]DADLE (delta) binding to 82.4 +/- 8.0% of the control value. (1S,2S)-trans-4-Isothiocyanato-N-methyl-N-[2-(1-pyrrolidinyl)- cyclohexyl]benzeneacetamide [(-)-3] exhibited selective wash-resistant inhibition of delta receptors labeled by [3H]DADLE resulting in a reduction in binding to 42.9 +/- 4.2% of control. In the alkyl isothiocyanate series, (1S,2S)-trans-N-methyl-N- [ 2- (1-pyrrolidinyl)cyclohexyl]-(S)-2-phenyl-2-isothiocyanatoacetamide [ (-)-11] also showed the capacity to selectively inhibit [3H]-U69,593-sensitive K sites, resulting in a reduction in binding to 72.2 * 2.54% of control at 1 pM while (+)-11 was inactive. None of the amino precursors (-)-8, (+)-8, (4-9, (+)-9, (-)-10, (+)-10, (-)-15,(+)-15, (-)-16, and (+)-16 of the isothiocyanates exhibited the capacity for wash-resistant inhibition of any of the receptor systems tested. Although intracerebroventricular (icv) injection of the most potent compound (-)-l into guinea pig brain failed to produce any irreversible inhibition of K receptors, icv injection of the less potent (-)-2into guinea pig brain resulted in a significant reduction in the K receptors that bind [3H]-U69,593, but not those that bind [3H]bremazocine. All of the compounds that showed the capacity to irreversibly inhibit K receptors labeled by [3H]-U69,593 in vitro possessed the 1S,2S absolute configuration.[2]
References
[1] LI Zm. Design and synthesis of high phase transition temperature compounds based on N-methylcyclohexylamine ligands and related properties[D].Jiangsu University of Science and Technology,2024.DOI:10.27171/d.cnki.ghdcc.2024.000080.
[2] de Costa BR, Rothman RB, Bykov V, et al. Probes for narcotic receptor mediated phenomena. 17. Synthesis and evaluation of a series of trans-3,4-dichloro-N-methyl-N-[2-(1-pyrrolidinyl)cyclohexyl]benzeneacet amide (U50,488) related isothiocyanate derivatives as opioid receptor affinity ligands. J Med Chem. 1990;33(4):1171-1176. doi:10.1021/jm00166a014
You may like
Related articles And Qustion
Lastest Price from N-Methylcyclohexylamine manufacturers

US $10.00/kg2025-04-21
- CAS:
- 100-60-7
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 100 mt

US $1190.00/kg2025-04-21
- CAS:
- 100-60-7
- Min. Order:
- 850kg
- Purity:
- 98%
- Supply Ability:
- 20 tons