Application research of 2-ethylhexyl 4-dimethylaminobenzoate
Introduction
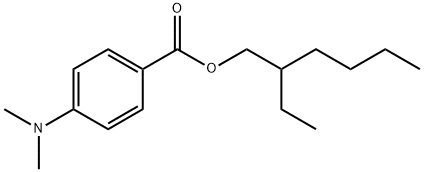
2-Ethylhexyl-4-dimethylaminobenzoate(Figure 1) is one of the more commonly used organic UV filters, and it has been detected in different water environments, sediments and aquatic organisms. 2-Ethylhexyl-4-dimethylaminobenzoate was detected in 100% of water samples in Qinhuai River, Jinchuan River, Xuanwu Lake and Yueya Lake within the vicinity of Nanjing City, China, with the concentrations ranging from 3 to 104 ng/L.The maximum concentration of 2-ethylhexyl-4-dimethylaminobenzoate reached 110 ng/L in tap water. 2-Ethylhexyl-4-dimethylaminobenzoate was detected at 150 µg/kg in the sediments of Hong Kong in Victoria Harbour. The highest concentration of 2-ethylhexyl-4-dimethylaminobenzoate in marine organisms from Hong Kong coastal waters was 24.1 µg/kg. Since the logarithm of the octanol/water partition coefficient (logKow) of this chemical is as high as 6.15, 2-ethylhexyl-4-dimethylaminobenzoate in water tends to adsorb onto sediments, and the logarithmic values of the organic carbon-normalized distribution coefficients (logKoc) were obtained and ranged from 4.37 to 4.44 in a lab-scale water-sediment system set up with natural water and sediment samples collected from Nanjing, East China. 2-Ethylhexyl-4-dimethylaminobenzoate can also be found to concentrate in fish by waterborne exposure and induce oxidative stress.[1]

Bioaccumulation and Biomagnification of 2-Ethylhexyl-4-dimethylaminobenzoate in Aquatic Animals.
The bioaccumulation and biomagnification of 2-ethylhexyl-4-dimethylaminobenzoate were investigated in two aquatic animals, the larvae of midge ( Chironomus riparius) and crucian carp ( Carassius carassius), and the metabolic enzyme responses in fish liver were determined. 2-Ethylhexyl-4-dimethylaminobenzoate in the larvae of midge reached a steady state within 10 days of sediment exposure. The biota-sediment accumulation factors ranged from 0.10 to 0.54, and were inversely proportional to the exposure concentrations. The 2-ethylhexyl-4-dimethylaminobenzoate-contaminated larvae were used to feed the crucian carp. Within 28 days of feeding exposure, the 2-ethylhexyl-4-dimethylaminobenzoate levels in fish tissues gradually increased with the increase of the exposure concentration, exhibiting an apparent concentration-dependence and time-dependence. The liver and kidneys were the main organs of accumulation, and the biomagnification factors of 2-ethylhexyl-4-dimethylaminobenzoate ranged from 8.97 to 11.0 and 6.44 to 10.8, respectively. In addition, 2-ethylhexyl-4-dimethylaminobenzoate significantly increased the activities of cytochrome P450 (CYP) 1A, CYP3A and glutathione S-transferase in the fish liver. Our results indicate that 2-ethylhexyl-4-dimethylaminobenzoate may pose a risk of biomagnification in an aquatic environment and influence the biological processes of exposed organisms.[1]
A direct observation of nonradiative dynamics from 2-ethylhexyl 4-dimethylaminobenzoate sunscreen agent.
Apart from being an analogue of the prototype for photoinduced intramolecular charge transfer (ICT), 2-ethylhexyl 4-dimethylaminobenzoate (EHDMABA) is also one of the earliest patented and most commonly used sunscreen components. There is, however, little documented information about the photophysics and factors affecting the photophysics of this molecule. Such information is of importance for both the understanding of the ICT reaction and assessing the underlying process of photoprotection, especially in view of the "sunscreen controversy" that has arisen from the contrasting in vivo vs. in vitro photobiological results on this and related UV filters. Researchers report herein a femtosecond broadband time-resolved fluorescence (fs-TRF), complemented by transient absorption (fs-TA) to allow a full probe of the excited state cascades for 2-ethylhexyl 4-dimethylaminobenzoate and two of its derivatives in solvents of varied properties. The results provide direct evidence for a nearly solvent independent inner sphere ICT reaction occurring on the sub-picosecond time scale, and an ensuing solvent dictated deactivation of the ICT state. The ICT state in the aprotic solvent acetonitrile decayed solely through the intrinsic intersystem crossing (ISC) to produce a potentially harmful triplet excited state. In the protic solvent, the solvation and formation of ICT-induced solute-solvent hydrogen (H)-bonding opened the originally inaccessible internal conversion (IC) channel of the ICT state, leading to the rapid reformation of the ground state molecule with a unitary efficiency in the aqueous solution. This H-bonding-mediated IC restrained or eliminated the intrinsic ISC, providing a mechanism at the molecular level for the benign dissipation of the electronic excitation. The precise rate of IC was observed to vary with the alkoxy substituent and its efficiency was affected by the H-bonding capacity of the solvent. The findings of this work demonstrate the pivotal role of the microenvironment and the direct participation of solvent molecules through H-bonding in drastically altering the nonradiative dynamics and promoting or inhibiting photostability and photoprotection. This may assist in developing next-generation UV filters and help in improving formulation design for the optimal efficacy of sunscreen products. The pronounced H-bonding-induced fluorescence quenching and variation in the fluorescence wavelength imply that these molecules may also serve as a sensitive fluorescence probe for the H-bonding properties of the microenvironment.[2]
The endocrine-disrupting effects of homosalate (HMS) and 2-ethylhexyl 4-dimethylaminobenzoate in rat pups
Homosalate (HMS) and 2-ethylhexyl 4-dimethylaminobenzoate (OD-PABA) are ultraviolet filters. Researchers aimed to investigate the effects of dermal exposure to HMS and 2-ethylhexyl 4-dimethylaminobenzoate during the prenatal, lactation, and early infancy periods on pubertal development and thyroid function in male and female rats. The thyroid glands, uteri, testes, prostate glands, and seminal vesicles were excised and weighed, the reproductive organs were analyzed histologically, and the serum hormone levels were measured. In the prenatal period, the thyroxine (T4) levels increased in the female rats in the exposed groups (p<0.05); the thyroid weights, reproductive organ weights, and gonadal hormone levels were not altered. In males, the testosterone levels decreased (p<0.05), but the thyroid weights, T4 levels, prostate, and testis weights were not changed. In the lactation period, the weights of the thyroid glands increased in the exposed female groups (p<0.05), but the T4, gonadal hormone levels, and reproductive organ weights were not changed. In the males, the thyroid gland weights, T4 levels, reproductive organ weights, and gonadal hormone levels were not changed. During infancy, the thyroid gland weights increased in the female rats in the exposed groups (p<0.05), but the T4 levels, gonadal hormone levels, and reproductive organ weights were not affected. In the male rats in the exposed groups, the T4 levels were increased (p<0.05), but the thyroid and reproductive organ weights, gonadal hormone levels were not affected. Organ histopathology was not affected in all groups. HMS and 2-ethylhexyl 4-dimethylaminobenzoate do not have endocrine disruptor effects on thyroid function and the pubertal development of female and male rats.[3]
References
[1]Lu G, Zhou R, Li S, Dang T, Liu J. Bioaccumulation and Biomagnification of 2-Ethylhexyl-4-dimethylaminobenzoate in Aquatic Animals. Int J Environ Res Public Health. 2018;15(11):2395. Published 2018 Oct 29. doi:10.3390/ijerph15112395
[2]Ma C , Chan CT , Chan RC , Wong AK , Chung BP , Kwok WM . Photoprotection or photodamage: a direct observation of nonradiative dynamics from 2-ethylhexyl 4-dimethylaminobenzoate sunscreen agent. Phys Chem Chem Phys. 2018;20(38):24796-24806. doi:10.1039/c8cp04447c
[3]Erol M, ?ok I, Bostan Gayret ?, et al. Evaluation of the endocrine-disrupting effects of homosalate (HMS) and 2-ethylhexyl 4-dimethylaminobenzoate (OD-PABA) in rat pups during the prenatal, lactation, and early postnatal periods. Toxicol Ind Health. 2017;33(10):775-791. doi:10.1177/0748233717718974
You may like
See also
Lastest Price from 2-Ethylhexyl 4-dimethylaminobenzoate manufacturers
US $0.00/kg2025-09-08
- CAS:
- 21245-02-3
- Min. Order:
- 1kg
- Purity:
- 99%min
- Supply Ability:
- 20tons

US $0.00-0.00/kg2025-06-11
- CAS:
- 21245-02-3
- Min. Order:
- 0.001kg
- Purity:
- 99.99%
- Supply Ability:
- 200000T