Application research of 3-Pyridinecarboxaldehyde
Introduction
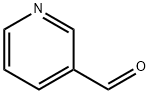
3-Pyridinecarboxaldehyde, also known as nicotinaldehyde (Figure 1), is an important heterocyclic aldehyde compound with significant applications in organic synthesis, pharmaceutical chemistry, and materials science.

The Conformational and Electronic Landscape of 3-Pyridinecarboxaldehyde
4-Pyridinecarboxaldehyde (3-PCA) is a versatile building block, but its conformational and ionization behavior remains underexplored. Using high-resolution IR-resonant vacuum ultraviolet mass-analyzed threshold ionization spectroscopy and Franck-Condon simulations, two conformers, s-cis and s-trans, were identified in the gas phase, distinguished by adiabatic ionization energies of 76123 ± 4 and 76173±4 cm-1, respectively, and revealing conformer-specific structural dynamics and frontier orbital changes during ionization. Computational analyses, including anharmonic and natural bond orbital studies, provided structural and electronic insights. Analysis of jet-cooled conformer populations indicated an S0-state energy difference of 40 ± 14 cm-1, while in the D0 state, the two conformers are essentially near-isoenergetic within experimental uncertainty (≈ -10±14 cm-1). These results highlight the delicate balance of cationic conformer stability and its distinct ionization behavior. This work provides conformer-specific spectroscopic and theoretical benchmarks for understanding ionization-induced structural and stereoelectronic effects in formyl-substituted pyridines with implications for their reactivity and excited-state dynamics.[1]
Direct coupling reaction of hydroquinone with 3-pyridinecarboxaldehyde
Synthesis of a novel dual inhibitor of thromboxane A2 synthetase and 5-lipoxygenase, 5,7-dimethyl-6-hydroxy-2-methylamino-4-(3- pyridylmethyl)benzothiazole (E3040), was accomplished via a new coupling reaction, in which a key intermediate, (3,6-dihydroxy-2,4-dimethylphenyl)-(3-pyridyl)methanol, was easily synthesized in a high yield from 2,6-dimethyl-1,4-benzohydroquinone and 3-pyridinecarboxaldehyde in 6 N hydrochloric acid. The regio isomers of 3-pyridinecarboxaldehyde also gave the corresponding coupling products in high yields.[2]
2,1'-dihydropyridomycins Synthesis
Dihydropyridomycins 2 and 3, which lack the characteristic enol ester moiety of the potent antimycobacterial natural product pyridomycin (1), have been prepared from l-Thr, R- and S-hydroxy isovaleric acid, and 3-pyridinecarboxaldehyde. The 2R isomer 2 shows only 4-fold lower anti-Mtb activity than 1, indicating that the enol ester moiety in the natural product is not critical for its biological activity. This finding establishes 2 as a potent and more practical lead for anti-TB drug discovery[3]
Inhibition of xanthine oxidase by various aldehydes
The inactivation of bovine milk xanthine oxidase by various aldehydes has been investigated. For each aldehyde, the inactivation reaction gives rise to a unique molybdenum(V) electron paramagnetic resonance signal from xanthine oxidase (the Inhibited signal). Of the aldehydes tested, only a few (mainly aromatic) failed to undergo this reaction. The g values of the Inhibited signals vary systematically from one aldehyde to another. As the substituents of the alpha-carbon atom become more electron withdrawing, so the gav increases. The inactivation rate depends on both enzyme and aldehyde concentration. Oxygen or another oxidizing substrate is also required for inhibition by 3-pyridinecarboxaldehyde and butyraldehyde but not formaldehyde. Reactivation of xanthine oxidase inhibited by an aldehyde occurs spontaneously after removal of excess aldehyde. For butyraldehyde or 3-pyridinecarboxaldehyde, greater than 95% recovery of activity was observed. The rate of reactivation is dependent both on the nature of the molecule bearing the aldehyde group and on a pK (6.6) of the complex with the enzyme. Evidence is presented that the modifying aldehyde in the Inhibited signal-giving species has (contrary to earlier assumptions) not been oxidized. These results are discussed in relation to the structure of the molybdenum center, and a mechanism for the inhibiting reaction is suggested.[4]
3-Pyridinecarboxaldehyde as an efficient PB reagent
Unsaturated fatty acids (UFAs) are fatty acids with one or more carbon-carbon double bonds (C=C). They are building blocks of structurally complex lipids. According to the position of C=C bond in the UFAs, there are multiple isomers with different physiological activities. Therefore, an accurate understanding of UFAs is essential. Combining Paternò-Büchi (PB) reaction with tandem mass spectrometry (MS/MS) analysis has been successfully used to identify unsaturated lipids in bio-samples. However, there are still some challenges, such as low reactive rate and low MS response of PB products. In this study, 3-pyridinecarboxaldehyde (3-PYA) was selected as an efficient PB reagent for the first time. According to the nitrogen rule, introduction of nitrogen did not only make the target MS and MS/MS ions to be easily identified but also increase the ionization efficiency significantly. The developed method in this study demonstrated greater efficiency and sensitivity for the identification of UFAs in total lipid extracts from bovine liver compared with other commonly used PB reagents.[5]
References
[1] Park SM, Kim H, Kwon CH. Unraveling the Conformational and Electronic Landscape of 3‑Pyridinecarboxaldehyde: A Spectroscopic Journey through Conformer-Specific Ionization. ACS Phys Chem Au. 2025;5(6):729-739. Published 2025 Oct 12. doi:10.1021/acsphyschemau.5c00088
[2] Komatsu Y, Minami N. Synthesis of a novel dual inhibitor of thromboxane A2 synthetase and 5-lipoxygenase (E3040) via the direct coupling reaction of hydroquinone with 3-pyridinecarboxaldehyde. Chem Pharm Bull (Tokyo). 1995;43(10):1614-1616. doi:10.1248/cpb.43.1614
[3] Horlacher OP, Hartkoorn RC, Cole ST, Altmann KH. Synthesis and antimycobacterial activity of 2,1'-dihydropyridomycins. ACS Med Chem Lett. 2012;4(2):264-268. Published 2012 Dec 18. doi:10.1021/ml300385q
[4] Morpeth FF, Bray RC. Inhibition of xanthine oxidase by various aldehydes. Biochemistry. 1984;23(6):1332-1338. doi:10.1021/bi00301a047
[5] Mao R, Li W, Jia P, et al. An efficient and sensitive method on the identification of unsaturated fatty acids in biosamples: Total lipid extract from bovine liver as a case study. J Chromatogr A. 2022;1675:463176. doi:10.1016/j.chroma.2022.463176
You may like
See also
Lastest Price from 3-Pyridinecarboxaldehyde manufacturers

US $45.00/kg2025-04-21
- CAS:
- 500-22-1
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20 tons

US $0.00/KG2025-04-02
- CAS:
- 500-22-1
- Min. Order:
- 1KG
- Purity:
- 98%
- Supply Ability:
- 2mt per month