Application Research of 4-Chloroformylphthalic anhydride
Introduction
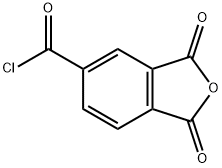
Trimellitic anhydride chloride (also known as 4-Chloroformylphthalic anhydride, Figure 1) is a highly reactive aromatic carboxylic acid derivative and a pivotal industrial chemical intermediate. Carboxylic anhydrides, one kind of carboxylic acid derivatives, have been extensively used in the field of epoxy curing system, amide coupling reaction and copolymer synthesis and so on. Analogous to acid chlorides, acid anhydrides with relatively low reactive activity could also be used as acylating agents for the amide bond formation through reaction with amines.[1]
Synthesis of polyamide (PA) composite membranes with tunable pore size and perm-selectivity
In the late 1970s, Cadotte et al. pioneered the fabrication of polyamide (PA) thin film composite (TFC) membranes through interfacial polymerization (IP) reaction, which has been the most important breakthrough in the developments of preparation technology of polymeric membranes. Because of high permeability, high selectivity, excellent robustness and durability in a broad range of pH (2−11) and temperature (0–45 °C), PA-TFC membranes have been widely utilized in many processes such as desalination, wastewater treatment, water purification and industrial substances separation.Generally, the PA selective separation layer is polymerized by diamines (aqueous monomers) and acid chlorides (organic monomers) on the organic phase side near the interface, which is attributed to the low solubility of acyl chlorides in the aqueous phase. Over the past decades, most studies have employed m-phenylenediamine (MPD) or piperazine (PIP) as the diamine monomer to react with trimesoyl chloride (TMC), which are the two most successful commercial membrane known as reverse osmosis (RO) and nanfiltration (NF) membrane, respectively.[1]Membranes with suitable nanopores and tunable charge characteristics are highly desired for their good application prospects in specific membrane-based separations. This study focused on the synthesis of polyamide (PA) composite membranes with tunable pore size and perm-selectivity. Trimellitic anhydride chloride (TAC) with an anhydride group, was introduced as the co-reactant of trimesoyl chloride (TMC) to form PA film via interfacial polymerization with m-phenylenediamine on the surface of the polysulfone substrate membrane. Due to the chemistry character difference (the number of functional group and acylation activity) between TMC and trimellitic anhydride chloride, the structure and physico-chemistry properties of the PA film could be tailored through the control of trimellitic anhydride chloride concentration. As a result, the nanopore radius of the membrane varied from <0.32 to 1.14 nm, along with the improved water permeability from 2.3 to 9.8 L/m2/h/bar and selectivity of Na2SO4/NaCl from 2.2 to 4.9. This study provided a facile and effective approach for the fabrication of tailor-made membrane with tunable nanopore and ion selectivity designed at a molecular level.[1]
Preparation of poly(amideimide siloxane)
A novel poly(amideimide siloxane) block copolymer was prepared from oligo(dimethylsiloxane), trimellitic anhydride chloride and oxylene diamine using trimethylsilyl chloride as the reaction activator. Trimethylsilyl chloride enhanced the reactivity of oligo(dimethylsiloxane), resulting in a higher yield and viscosity of the product. The high reactivity of N-trimethylsilylated siloxane diamine stems from the silicon σ—π effect, and a nucleophilic addition—elimination reaction is proposed. The addition of a small amount of N-methylpyrrolidone in tetrahydrofuran led to a higher yield. The temperature of 10% weight loss for the product exceeded 355°C according to thermogravimetric analysis data. A transient change in the contact angle of water on the sample surface was observed, suggesting surface segregation of the hydrophobic component in the polymer.[2]
Preparation and characterization of an amphoteric chitosan derivative
Few modified chitosan based films have been developed for drug delivery applications mainly due to difficulty in dissolving modified chitosan matrices or requirement of acidic media in order to prepare the films. The main potential for drug delivery using chitosan films logically lies with topical applications. For example, chitosan films have been studied for theirpotential for topical drug (ondasetron) release, wound dressing and artificial skin. Trimellitic anhydride chloride was used to generate a water soluble amphoteric chitosan derivative (CTAA). CTAA was combined with alginate to facilitate direct targeting of 5-FU to the colon. The structure of CTAA was characterized by FTIR, 1H/13C NMR and HSQC. The physical properties of the CTAA hydrogel were analyzed by scanning electron microscopy, X-ray diffraction and thermogravimetry. Inhibition of a pancreatic enzyme's activity and cytotoxicity studies of CTAA were also carried out. Sodium alginate was mixed with CTAA to form a polyelectrolyte complex film and resulted in improved controlled drug release. Swelling characteristics of the films as a function of pH were also investigated. An optimized formulation was exposed to conditions simulating the stomach, small intestine, and colon. The CTAA/alginate formulation used in this preliminary study presents potential for at least 28% of ingested 5-FU to arrive at the colon ready for release by colonic enzymes. Films were chosen in the study to severely stretch the capabilities of the CTAA/alginate system. The system nevertheless will need modifying for in vivo delivery to the colon.[3]
References
[1] Zhang Z , Kang G , Yu H ,et al.From reverse osmosis to nanofiltration: Precise control of the pore size and charge of polyamide membranes via interfacial polymerization[J].Desalination, 2019, 466:16-23.DOI:10.1016/j.desal.2019.05.001.
[2] Seong Yong Ha,Boo-Keun Oh,Young Moo Lee.Preparation of poly(amideimide siloxane) from trimellitic anhydride chloride, oxylene diamine and oligo(dimethylsiloxane) diamine[J].Polymer, 1995.DOI:10.1016/0032-3861(95)92027-C.
[3] Kavianinia I , Plieger P G , Kandile N G ,et al.Preparation and characterization of an amphoteric chitosan derivative employing trimellitic anhydride chloride and its potential for colon targeted drug delivery system[J].Materials Today Communications, 2015, 3:78-86.DOI:10.1016/j.mtcomm.2015.03.002.
You may like
See also
Lastest Price from 4-Chloroformylphthalic anhydride manufacturers

US $0.00/kg2025-03-07
- CAS:
- 1204-28-0
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 1000kg

US $25.00/KG2023-03-27
- CAS:
- 1204-28-0
- Min. Order:
- 1KG
- Purity:
- 0.99
- Supply Ability:
- 50000KG/MONTH