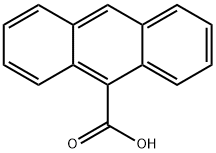
Application Research of 9-anthracenecarboxylic acid
Introduction
The chloride-channel inhibitor 9-anthracenecarboxylic acid (9-AC;Figure 1) specifically blocks the skeletal muscle-type chloride channel ClC-1. Malfunction of this channel,essential for the stability of the resting membrane potential in skeletal muscle , causes myotonia, a pathological condition also induced by 9-anthracenecarboxylic acid. ClC-1 channels are also present in outer hair cells (OHCs) of the cochlea, where chloride ions are essential for normal hearing.Patients suffering from myotonic dystrophy also have a concurrent high risk of sensorineural hearing loss.[1] 9-Anthracenecarboxylic acid had a significant inhibitory effect on the proliferation of human myeloid leukemia cell line HL-60, but its inhibitory activity on other solid tumor cells has not been reported.

Cyclometalated iridium (III) complex based on 9-Anthracenecarboxylic acid
9-Anthracenecarboxylic acid (9-Ac) was reported early as a chloride channel inhibitor and was found to exhibit significant anti-proliferative activity on leukemic cells, but has not been researched in solid tumor cells. Herein, a 9-anthraceneic acid derivative was introduced into the cyclometalated Iridium (III) species to construct a novel Iridium (Ir) complex Ir-9-Ac, [Ir(ppy) 2(9-Ac-L)]PF 6 (ppy = 2-phenylpyridine, 9-Ac-L = N-((4'-methyl-[2,2'-bipyridin]-4-yl)methyl)anthracene-9-carboxamide), which could accumulated in lysosomes. Ir-9-Ac showed good cytotoxic activity against several tumor cell lines, notably on A549 cells. Besides Ir-9-Ac could inhibit the cell colony formation and growth of the 3D cell spheroids, demonstrating the potential to suppress tumors in vivo. This design provided a platform for the design of cyclometalated Iridium (III) anticancer complexes.[2]
Reduces the nonlinear capacitance of prestin-associated charge movement
9-Anthracenecarboxylic acid, the chloride channel blocker used in the reticular lamina experiments,has also been shown to reduce the magnitude of the imaginary part of the OHC axial mechanical impedance. Although the mechanical impedance data suggest that the axial stiffness of the cell was reduced by 9-anthracenecarboxylic acid, the mechanisms of 9-anthracenecarboxylic acid block are unknown. Given the importance of the chloride sensitivity of prestin and of putative links between hearing loss and chloride-associated channelopathies, such as in myotonic dystrophy, the aim of the present study was to elucidate mechanisms of 9-anthracenecarboxylic acid block of OHC function.
To elucidate these 9-anthracenecarboxylic acid effects, the functional electromechanical status of prestin was assayed by measuring the nonlinear capacitance of OHCs from the guinea-pig cochlea and of prestin-transfected human embryonic kidney 293 (HEK 293) cells. Extracellular application of 9-anthracenecarboxylic acid caused reversible, dose-dependent and chloride-sensitive reduction in OHC nonlinear charge transfer, Qmax . Prestin-transfected cells also showed reversible reduction in Qmax . For OHCs, intracellular 9-anthracenecarboxylic acid application as well as reduced intracellular pH had no detectable effect on the reduction in Qmax by extracellularly applied 9-anthracenecarboxylic acid. In the prestin-transfected cells, cytosolic application of 9-anthracenecarboxylic acid approximately halved the blocking efficacy of extracellularly applied 9-anthracenecarboxylic acid. OHC inside-out patches presented the whole-cell blocking characteristics. Disruption of the cytoskeleton by preventing actin polymerization with latrunculin A or by decoupling of spectrin from actin with diamide did not affect the 9-AC-evoked reduction in Qmax . We conclude that 9-anthracenecarboxylic acid acts on the electromechanical transducer principally by interaction with prestin rather than acting via the cytoskeleton, chloride channels or pH. The 9-anthracenecarboxylic acid block presents characteristics in common with salicylate, but is almost an order of magnitude faster. 9-Anthracenecarboxylic acid provides a new tool for elucidating the molecular dynamics of prestin function.[1]
Spectrofluorimetric determination of 5-fluorouracil by fluorescence quenching
Photo-induced intermolecular electron transfer (PET) interaction between excited singlet (S(1)) state of 9-anthracenecarboxylic acid and DNA bases of pyrimidines as uracil and 5-fluorouracil (5-FU) has been studied in water and ethanol solutions using steady-state fluorescence spectroscopy. The intensity of all emission bands of 9-anthracenecarboxylic acid was quenched in presence of uracil and 5-FU by electron transfer reaction without formation of an exciplex. It was found that uracil and 5-fluorouracil acts as effective electron donors and simultaneously quench the fluorescence of electron-accepting sensitizer 9-anthracenecarboxylic acid. The quenching by diffusion-controlled rate coincides well with the dynamic Stern-Volmer correlation. The bimolecular quenching rate constant (k(q)(ss)) and electron transfer rate constant (k(et)) observed are seen to be much higher for 5-fluorouracil than those for uracil. The thermodynamic parameters estimated by using the Rehm-Weller equation were used to propose a suitable mechanism for PET occurring between uracils and 9-anthracenecarboxylic acid. The proposed method was used to determine 5-fluorouracil from pharmaceutical samples with satisfactory results. The technique is more selective, sensitive and relatively free from coexisting substances.[3]
A new zinc complex with 9-Anthracenecarboxylic Acid Synthesis
A new zinc complex, [Zn (9-AC)2] (1) (9-AC=9-anthracenecarboxylic acid), was prepared via conventional electrochemical method in a fast and facile process and fully characterized by 1H-NMR, 13C-NMR, IR spectroscopy and elemental analysis. The nano structures of the same compound were successfully produced by a facile and environment-friendly sonoelectrochemical route at different current densities (0.5, 1.2, 1.8, 2.5 and 3.5 mA/cm2). The new nano-structure particles were characterized by scanning electron microscopy, X-ray powder diffraction, IR spectroscopy and elemental analysis. Thermal stability of single crystal and nano-size samples of the prepared compound was studied by thermogravimetric and differential thermal analysis. The comparison of the effect of current density without and with ultrasonic irradiation on particle size has been investigated in convectional electrochemical and sonoelectrochemical method respectively. The results showed that using ultrasonic irradiation with increasing the current density lead to decrease the particle sizes unlike conventional electrochemical method. In other words, when the current density increase from 0.5 to 3.5 mA/cm2, in sonoelectrochemical method, the particle sizes decrease from 100 to 48 nm while, in convectional electrochemical method, the particle sizes increase from 400 to 1200 nm and possible explanation offered. Photoluminescence properties of the nano-structured and crystalline bulk of the prepared complex at room temperature in the solid state have been investigated in detail. The results indicate that the size of the complex particles has an important effect on their optical properties.[4]
References
[1] Harasztosi C, Gummer AW. The chloride-channel blocker 9-anthracenecarboxylic acid reduces the nonlinear capacitance of prestin-associated charge movement. Eur J Neurosci. 2016;43(8):1062-1074. doi:10.1111/ejn.13209
[2] Liu L, Chen J, Wang MM, et al. The cyclometalated iridium (III) complex based on 9-Anthracenecarboxylic acid as a lysosomal-targeted anticancer agent. J Inorg Biochem. 2022;235:111913. doi:10.1016/j.jinorgbio.2022.111913
[3] Khot MS, Bhattar SL, Kolekar GB, Patil SR. Spectrofluorimetric determination of 5-fluorouracil by fluorescence quenching of 9-anthracenecarboxylic acid. Spectrochim Acta A Mol Biomol Spectrosc. 2010;77(1):82-86. doi:10.1016/j.saa.2010.04.029
[4] Shahrjerdi A, Davarani SS. Sonoelectrochemical Synthesis of Nano Zinc (II) Complexes with 9-Anthracenecarboxylic Acid: Effect of Current Density and Study of their Photophysical Properties. J Fluoresc. 2016;26(6):2053-2061. doi:10.1007/s10895-016-1900-6
You may like
See also
Lastest Price from 9-Anthracenecarboxylic acid manufacturers

US $0.00/kg2026-03-25
- CAS:
- 723-62-6
- Min. Order:
- 1kg
- Purity:
- 99%min
- Supply Ability:
- 20tons

US $0.00/kg2025-11-28
- CAS:
- 723-62-6
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 1kg 25kg 50kg 100kg 1000kg