Application Research of Cerium(iii) nitrate hexahydrate
Introduction
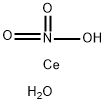
Cerium(III) nitrate hexahydrate is an inorganic compound with a white crystalline appearance (Figure 1). Its main applications include serving as an analytical chemistry reagent, a catalyst in the petrochemical industry, an additive for gas lamp mantles, a raw material for cerium salt preparation, as well as in the pharmaceutical industry and the production of tungsten-molybdenum products (e.g., cerium-tungsten electrodes). In the field of energy storage materials, the synergistic catalysis of cerium nitrate and lithium hydroxide can significantly enhance the thermal energy storage performance of magnesium hydroxide. This paper mainly enumerates the relevant application research thereof.

Antimicrobial Activity of Citrate-Coated Cerium Oxide Nanoparticles
The purpose of this study was to investigate the antimicrobial activity of citrate-stabilized sols of cerium oxide nanoparticles at different concentrations via different microbiological methods and to compare the effect with the peroxidase activity of nanoceria for the subsequent development of a regeneration-stimulating medical and/or veterinary wound-healing product providing new types of antimicrobial action. The object of this study was cerium oxide nanoparticles synthesized from aqueous solutions of cerium (III) nitrate hexahydrate and citric acid (the size of the nanoparticles was 3-5 nm, and their aggregates were 60-130 nm). Nanoceria oxide sols with a wide range of concentrations (10-1-10-6 M) as well as powder (the dry substance) were used. Both bacterial and fungal strains (Bacillus subtilis, Bacillus cereus, Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, Proteus vulgaris, Candida albicans, Aspergillus brasielensis) were used for the microbiological studies. The antimicrobial activity of nanoceria was investigated across a wide range of concentrations using three methods sequentially; the antimicrobial activity was studied by examining diffusion into agar, the serial dilution method was used to detect the minimum inhibitory and bactericidal concentrations, and, finally, gas chromatography with mass-selective detection was performed to study the inhibition of E. coli's growth. To study the redox activity of different concentrations of nanocerium, we studied the intensity of chemiluminescence in the oxidation reaction of luminol in the presence of hydrogen peroxide. As a result of this study's use of the agar diffusion and serial dilution methods followed by sowing, no significant evidence of antimicrobial activity was found. At the same time, in the current study of antimicrobial activity against E. coli strains using gas chromatography with mass spectrometry, the ability of nanoceria to significantly inhibit the growth and reproduction of microorganisms after 24 h and, in particular, after 48 h of incubation at a wide range of concentrations, 10-2-10-5 M (48-95% reduction in the number of microbes with a significant dose-dependent effect) was determined as the optimum concentration. A reliable redox activity of nanoceria coated with citrate was established, increasing in proportion to the concentration, confirming the oxidative mechanism of the action of nanoceria. Thus, nanoceria have a dose-dependent bacteriostatic effect, which is most pronounced at concentrations of 10-2-10-3 M. Unlike the effects of classical antiseptics, the effect was manifested from 2 days and increased during the observation. To study the antimicrobial activity of nanomaterials, it is advisable not to use classical qualitative and semi-quantitative methods; rather, the employment of more accurate quantitative methods is advised, in particular, gas chromatography-mass spectrometry, during several days of incubation.[1]
CuCeTA nanoflowers as an efficient peroxidase candidate
Conventional nanozyme-based pesticide detection often requires the assistance of acetylcholinesterase. In this work, a CuCeTA nanozyme was successfully designed for the direct colorimetric detection of glyphosate. Direct detection can effectively avoid the problems caused by cascading with natural enzymes such as acetylcholinesterase. By assembling tannic acid, copper sulfate pentahydrate and cerium(III) nitrate hexahydrate, CuCeTA nanoflowers were prepared. The obtained CuCeTA possessed excellent peroxidase-like activity that could catalyze the oxidation of 3,3',5,5'-tetramethylbenzidine (TMB) to blue oxidized TMB in the presence of hydrogen peroxide. Glyphosate could effectively inhibit the peroxidase-like activity of CuCeTA while other pesticides (fenthion, chlorpyrifos, profenofos, phosmet, bromoxynil and dichlorophen) did not show significant inhibitory effects on the catalytic activity of CuCeTA. In this way, CuCeTA could be used for the colorimetric detection of glyphosate with a low detection limit of 0.025 ppm. Combined with a smartphone and imageJ software, a glyphosate test paper was designed with a detection limit of 3.09 ppm. Fourier transform infrared spectroscopy demonstrated that glyphosate and CuCeTA might be bound by coordination, which could affect the catalytic activity of CuCeTA. These CuCeTA-based nanozyme system exhibited unique selectivity and sensitivity for glyphosate detection and this work may provide a new strategy for rapid and convenient detection of pesticides.[2]
Photocatalysis by CeO2-Loaded Soybean Powder Carbon
In the process of using photocatalysts to treat tetracycline (TC) wastewater, the degradation efficiency of soybean powder carbon material (SPC) can be improved by loading it with cerium oxide (CeO2). In this study, firstly, SPC was modified by phytic acid. Then, the CeO2 was deposited on modified SPC using the self-assembly method. Catalyzed cerium (III) nitrate hexahydrate (CeH3NO4) was treated with alkali and calcined at 600°C under nitrogen. XRD, XPS, SEM, EDS, UV-VIS /DRS, FTIR, PL and N2 adsorption-desorption methods were used to characterize the crystal structure, chemical composition, morphology, surface physical and chemical properties. The effects of catalyst dosage, monomer contrast, pH value and co-existing anions on TC oxidation degradation were investigated, and the reaction mechanism of a 600 Ce-SPC photocatalytic reaction system was discussed. The results show that the 600 Ce-SPC composite presents uneven gully morphology, which is similar to the natural "briquettes". The degradation efficiency of 600 Ce-SPC reached about 99% at 60 min under light irradiation when the optimal catalyst dosage and pH were 20 mg and 7. Meanwhile, the reusability of the 600 Ce-SPC samples showed good stability and catalytic activity after four cycles.[3]
Nitrates of cerium and samarium deposit on human enamel
The aim of this study was to analyze the precipitation of Cerium(III) nitrate hexahydrate or Samarium(III)nitrate hexahydrate [Sm(NO3)3] solutions on human enamel with and without a salivary pellicle. Investigated parameters were At%Ce and At%Sm measured using energy dispersive x-ray spectroscopy (EDX) after test solution (two concentrations) application.
Precipitation of Cerium(III) nitrate hexahydrate and Sm(NO3)3 solutions was examined on human enamel with and without a salivary pellicle. 6 enamel specimens each were obtained from 12 freshly extracted human third molars. These specimens were ground flat and polished. A salivary pellicle was created on 3 of the 6 specimens per tooth by storing the samples in human saliva. Subsequently, an aqueous solution of Cerium(III) nitrate hexahydrate was applied to 2 of the 6 specimens (one with, one without salivary pellicle) for 60 s. The same was carried out with an aqueous solution of Sm(NO3)3 on 2 further specimens. The remaining 2 specimens from each tooth were treated with demineralized water (negative control). Cerium(III) nitrate hexahydrate and Sm(NO3)3 solutions were applied at 25 or 50 wt% (aqueous solutions). The test materials and concentrations were distributed using a randomization table. After 60 s exposure and rinsing with demineralized water, the elemental composition (Ce, Sm, Ca, P, O, N, Na, Mg) of the enamel surface was analyzed by EDX. Atomic percentages (At%), differences (ΔAt%) and calcium/phosphorous-ratios (Ca/P-ratios) were calculated and analyzed non-parametrically (α = 0.05). 2.0-2.3 At%Ce (median) was detected on Cerium(III) nitrate hexahydrate-treated enamel and 0.4-0.7 At% Sm (median) was detected on Sm(NO3)3-treated enamel. Ce was only detected on the surfaces after application of Cerium(III) nitrate hexahydrate, Sm only after application of Sm(NO3)3. The Ca/P-ratio was significantly lower (1.37-1.59; p = 0.028) after the application of 25% and 50%Ce(NO3)3 as well as 50%Sm(NO3)3 compared to the control treatment (demineralized water; 1.61-1.63). After treatment with Cerium(III) nitrate hexahydrate, At%Ca and At%Na were significantly lower (p ≤ 0.043) compared to treatment with Sm(NO3)3. No significant differences were found between specimens treated with 25% or 50% lanthanide nitrate solution. Presence of a salivary pellicle had no significant influence on the measured At% with the exception of specimens treated with 50% Sm(NO3)3 with increased At%Sm (p ≤ 0.046). Cerium(III) nitrate hexahydrate and Sm(NO3)3 precipitate on human enamel independently of the presence of a salivary pellicle.[4]
References
[1] Silina EV, Ivanova OS, Manturova NE, et al. Antimicrobial Activity of Citrate-Coated Cerium Oxide Nanoparticles. Nanomaterials (Basel). 2024;14(4):354. Published 2024 Feb 13. doi:10.3390/nano14040354
[2] Jiang C, Zhong H, Zou J, Zhu G, Huang Y. CuCeTA nanoflowers as an efficient peroxidase candidate for direct colorimetric detection of glyphosate. J Mater Chem B. 2023;11(40):9630-9638. Published 2023 Oct 18. doi:10.1039/d3tb01455j
[3] He X, Qin W, Xie Y. Degradation of Tetracycline with Photocatalysis by CeO2-Loaded Soybean Powder Carbon. Nanomaterials (Basel). 2023;13(6):1076. Published 2023 Mar 16. doi:10.3390/nano13061076
[4] Kopp L, Hiller KA, Cieplik F, et al. Nitrates of cerium and samarium deposit on human enamel independently of a salivary pellicle. Front Oral Health. 2024;5:1455924. Published 2024 Aug 29. doi:10.3389/froh.2024.1455924
You may like
See also
Lastest Price from CERIUM(III) NITRATE HEXAHYDRATE manufacturers

US $0.00-0.00/KG2025-04-15
- CAS:
- 10294-41-4
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 500000kg

US $20.00-10.00/kg2025-03-07
- CAS:
- 10294-41-4
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 10 tons