Application research of Cocarboxylase
Introduction
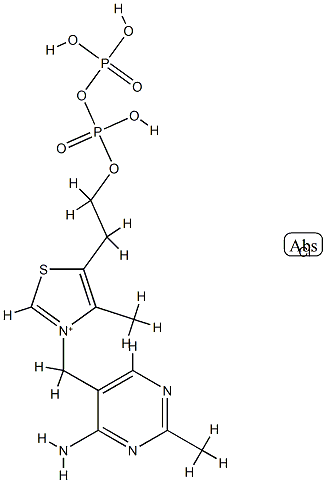
In recent years, due to the synthetic production of cocarboxylase, interest in its use in a number of diseases has increased. Cocarboxylase, also known as thiamine pyrophosphate (TPP;Figure 1),is a cofactor for 2-hydroxyacyl-CoA lyase 1 (HACL1), a peroxisomal enzyme essential for the alpha-oxidation of phytanic acid and 2-hydroxy straight chain fatty acids. So far, HACL1 is the only known peroxisomal TPP-dependent enzyme in mammals. Little is known about the transport of metabolites and cofactors across the peroxisomal membrane and no peroxisomal thiamine or TPP carrier has been identified in mammals yet.Because of the crucial role of the cofactor cocarboxylase, Fraccascia et al. reanalyzed its subcellular localization in rat liver. In addition to the known mitochondrial and cytosolic pools, we demonstrated, for the first time, that peroxisomes contain cocarboxylase (177 +/- 2 pmol/mg protein). Subsequently, we verified whether cocarboxylase could be synthesized from its precursor thiamine, in situ, by a peroxisomal thiamine pyrophosphokinase (TPK). However, TPK activity was exclusively recovered in the cytosol. These results clearly indicate that mammalian peroxisomes do contain cocarboxylase but that no pyrophosphorylation of thiamine occurs in these organelles, implying that thiamine must enter the peroxisome already pyrophosphorylated. Consequently, cocarboxylase entry may depend on a specific transport system or, in a bound form, on HACL1 translocation.[1]

Open-chain thiamine analogues as potent inhibitors of cocarboxylase-dependent enzymes
A common approach to studying cocarboxylase-dependent enzymes is by chemical inhibition with thiamine/cocarboxylase analogues which feature a neutral aromatic ring in place of the positive thiazolium ring of cocarboxylase. These are potent inhibitors but their preparation generally involves multiple synthetic steps to construct the central ring. Chan and his colleagues had reported efficient syntheses of novel, open-chain thiamine analogues which potently inhibit cocarboxylase-dependent enzymes and are predicted to share the same binding mode as cocarboxylase. They also report some open-chain analogues that inhibit pyruvate dehydrogenase E1-subunit (PDH E1) and are predicted to occupy additional pockets in the enzyme other than the TPP-binding pockets. This opens up new possibilities for increasing the affinity and selectivity of the analogues for PDH, which is an established anti-cancer target.[2]
Beneficial effects of cocarboxylase in the treatment of experimental myocardial infarction in dogs
Cocarboxylase, or thiamine pyrophosphate, is an essential coenzyme in the catabolism of pyruvate. The authors evaluated the effects of a stable cocarboxylase solution in the treatment of an experimentally created acute myocardial infarction in 14 healthy mongrel dogs. The left anterior descending artery was ligated for 60 minutes and data were collected at the following points: A) prior to ligation, B) 15 minutes after ligation, C) 30 minutes after ligation, and D) 60 minutes after ligation. In one group (Group II), cocarboxylase (150 mgm/kg) was given systematically via a central line 15 minutes and 45 minutes after ligation, while in Group I an equal amount of D5W was given. Hemodynamic data include heart rate, systolic and mean arterial pressure, pulmonary wedge pressure, right arterial pressure, and cardiac output. Myocardial O2 consumption was determined by the method of Rooke and Feigl. Electrocardiographic data were also monitored throughout the experiment. In both groups, preligation (point A) hemodynamic data were similar. In Group II, there were beneficial hemodynamic changes versus Group I (expressed as percentage recovery of hemodynamic performance from preligation) at points C and D, with significant (P less than 0.05) decreases in heart rate, increased stroke volume, decreased systemic vascular resistance, and decreased myocardial O2 consumption. EKG criteria also showed improvement in Group II versus Group I. In conclusion, this experiment suggests that cocarboxylase may be beneficial to ischemic canine myocardium by virtue of its favorable systemic hemodynamic effects.[3]
Use of cocarboxylase to prevent infertility developing in rats
To investigate whether cocarboxylasee can prevent infertility developing in rats undergoing unilateral ovariectomy and with ischemia reperfusion induced in the contralateral ovary. Biochemical examinations of the ovaries were also performed. Rats were divided into two main groups of three subgroups each. An ischemia reperfusion model was established in the first main group, while surgical unilateral ovariectomy was performed in the second. Cocarboxylase and melatonin were administered to the subgroups. No additional procedure was performed in the control groups. The rats were then left in laboratory environments and their fertility levels were determined. Malondialdehyde, total glutathione and DNA damage products were measured in those rats from which ovarian tissue was collected. The results showed that cocarboxylase prevented ischemia/reperfusion injury-related infertility, but melatonin did not provide adequate prevention. However, reproduction in healthy animals receiving melatonin began earlier compared to those receiving cocarboxylase. Melatonin suppressed oxidative stress caused by ischemia/reperfusion in ovarian tissue significantly better than did cocarboxylase. Yapca et al. think that different mechanisms, in addition to antioxidant activity, are involved in the prevention of reperfusion-associated infertility after ischemia.[4]
References
[1] Fraccascia P, Sniekers M, Casteels M, Van Veldhoven PP. Presence of thiamine pyrophosphate in mammalian peroxisomes. BMC Biochem. 2007;8:10. Published 2007 Jun 27. doi:10.1186/1471-2091-8-10
[2] Chan AHY, Ho TCS, Leeper FJ. Open-chain thiamine analogues as potent inhibitors of thiamine pyrophosphate (TPP)-dependent enzymes. Org Biomol Chem. 2023;21(32):6531-6536. Published 2023 Aug 16. doi:10.1039/d3ob00884c
[3] Larrieu AJ, Yazdanfar S, Redovan E, et al. Beneficial effects of cocarboxylase in the treatment of experimental myocardial infarction in dogs. Am Surg. 1987;53(12):721-725.
Yapca OE, Turan MI, Cetin N, Borekci B, Gul MA. Use of thiamine pyrophosphate to prevent infertility developing in rats undergoing unilateral ovariectomy and with ischemia reperfusion induced in the contralateral ovary. Eur J Obstet Gynecol Reprod Biol. 2013;170(2):521-525. doi:10.1016/j.ejogrb.2013.07.027You may like
Related articles And Qustion
Lastest Price from Cocarboxylase manufacturers
US $1.00-4.00/KG2025-09-11
- CAS:
- 154-87-0
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 200000KG

US $10.00/ASSAYS2025-08-20
- CAS:
- 154-87-0
- Min. Order:
- 1ASSAYS
- Purity:
- 99%
- Supply Ability:
- 1 ton