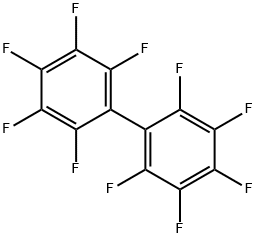
Application research of Decafluorobiphenyl
Introduction
Beside being important in (bio)molecules, small fluorinated molecules could be useful as linkers used for bioconjugation, a growing field in chemical biology and drug discovery and could be another way of introducing fluorine into organic molecules with pharmaceutical potential. Among the fluorine containing moieties, decafluorobiphenyl (DFBP; Figure 1) has raised interest in exhibiting high reactivity under simple SNAr reaction conditions. In addition, decafluorobiphenyl is chemically robust and commercially available. Schubert considers this system to satisfy many of the requirements of a “click” reaction, making it attractive for bioconjugation.

Considerable attention has been devoted to fluorinated compounds due to their unique and interesting properties. Many modern pharmaceuticals contain fluorinated substituents, which are commonly synthesized using selective fluorinating reagents. Decafluorobiphenyl as a fluorinated linker is susceptible to nucleophilic attack. This nucleophilic reaction has been widely studied using various nucleophiles. Sulfur and nitrogen containing nucleophiles have been of particular interest, especially in bioconjugated reactions. The SNAr reactivity of decafluorobiphenyl and its application in synthesising bioactive compounds were reported. The SNAr reaction on decafluorobiphenyl can be classified into three types: (1) C-S (2) C-N and (3) C-O. C-S bond formation was extensively used as a linker in peptide chemistry with C-N bond formation to a lesser extent. C-O bond formation of decafluorobiphenyl has also been studied by several groups for the preparation of poly(arylene sulfones) as conductive electronic devices.[1]
Development of a Decafluorobiphenyl Cyclized Peptide Targeting the NEMO-IKKα/β Interaction
Aberrant canonical NF-κB signaling has been implicated in diseases, such as autoimmune disorders and cancer. Direct disruption of the interaction of NEMO and IKKα/β has been developed as a novel way to inhibit the overactivation of NF-κB. Peptides are a potential solution for disrupting protein-protein interactions (PPIs); however, they typically suffer from poor stability in vivo and limited tissue penetration permeability, hampering their widespread use as new chemical biology tools and potential therapeutics. In this work, decafluorobiphenyl-cysteine SNAr chemistry, molecular modeling, and biological validation allowed the development of peptide PPI inhibitors. The resulting cyclic peptide specifically inhibited canonical NF-κB signaling in vitro and in vivo, and presented positive metabolic stability, anti-inflammatory effects, and low cytotoxicity. Importantly, our results also revealed that cyclic peptides had huge potential in acute lung injury (ALI) treatment, and confirmed the role of the decafluorobiphenyl-based cyclization strategy in enhancing the biological activity of peptide NEMO-IKKα/β inhibitors. Moreover, it provided a promising method for the development of peptide-PPI inhibitors.[2]
Quadruply charged decafluorobiphenyl
Femtosecond laser ionization is a unique means to produce multiply charged organic molecules in the gas phase. The charge-dependent chemical reactions of such electron-deficient molecules are interesting from both fundamental and applied scientific perspectives. Kitagawa et al. have reported the production of quadruply charged perfluoroaromatics; however, they were so stable that Kitagawa et al. cannot obtain information about their chemical reactions. In general, it might be difficult to realize the conflicting objectives of observing multiply charged molecular ion themselves and their metastable dissociations. In this study, Kitagawa et al. report the first example showing metastable dissociations of several charge states within the measurable time range of a time-of-flight mass spectrometer. Metastable dissociations were analyzed by selecting a precursor ion with a Bradbury-Nielsen ion gate followed by time-of-flight analysis using a reflectron. Kitagawa et al. obtained qualitative information that triply and quadruply charged decafluorobiphenyl survived at least in the acceleration region but completely decomposed before entering a reflectron. In contrast, three dissociation channels for singly and one for doubly charged molecular ions were discriminated by a reflectron and determined with the help of ion trajectory simulations.[3]
Cysteine-Based Perfluorinated Derivatives synthesized
Cysteine-based perfluoroaromatic (hexafluorobenzene (HFB) and decafluorobiphenyl (DFBP)) were synthesized and established as a chemoselective and available core to construct molecular systems ranging from small molecules to biomolecules with interesting properties. The decafluorobiphenyl was found more effective than HFB for the monoalkylation of decorated thiol molecules. As proof of concept of the potential application of perfluorinated derivatives as non-cleavable linkers, some antibody-perfluorinated conjugates were prepared via thiol through two different strategies, i) using thiol from reduced cystamine coupling to carboxylic acids from mAb by amide bond, and ii) using thiols from reduction of mAb disulfide bond. Conjugates cell binding analysis demonstrated that the bioconjugation does not affect the macromolecular entity. Besides, some molecular properties of synthesized compounds are evaluated through spectroscopic characterization (FTIR and 19F-NMR chemical shifts) and theoretical calculations. The comparison of calculated and experimental 19F-NMR shifts and IR wavenumbers give excellent correlations, asserting as powerful tools in structurally identifying HFB and decafluorobiphenyl derivatives. Moreover, molecular docking was also developed to predict cysteine-based perfluorated derivatives' affinity against topoisomerase Il and cyclooxygenase 2 (COX-2). The results suggested that mainly cysteine-based decafluorobiphenyl derivatives could be potential topoisomerase II α and COX-2 binders, becoming potential anticancer agents and candidates for anti-inflammatory treatment.[4]
References
[1] Alapour S, de la Torre BG, Ramjugernath D, Koorbanally NA, Albericio F. Application of Decafluorobiphenyl (DFBP) Moiety as a Linker in Bioconjugation. Bioconjug Chem. 2018;29(2):225-233. doi:10.1021/acs.bioconjchem.7b00800
[2] Li S, Song S, Liu X, et al. Development of a Decafluorobiphenyl Cyclized Peptide Targeting the NEMO-IKKα/β Interaction that Enhances Cell Penetration and Attenuates Lipopolysaccharide-Induced Acute Lung Injury. Bioconjug Chem. 2024;35(5):638-652. doi:10.1021/acs.bioconjchem.4c00122
[3] Kitagawa K, Fujihara A, Yatsuhashi T. Charge-Dependent Metastable Dissociations of Multiply Charged Decafluorobiphenyl Formed by Femtosecond Laser Pulses. Mass Spectrom (Tokyo). 2023;12(1):A0130. doi:10.5702/massspectrometry.A0130
[4] Chalán-Gualán S, Ramos-Tomillero I, Terencio T, et al. Cysteine-Based Perfluorinated Derivatives: A Theoretical and Experimental Study. Chempluschem. 2023;88(5):e202300028. doi:10.1002/cplu.202300028
You may like
See also
Lastest Price from Decafluorobiphenyl manufacturers
US $0.00-0.00/kg2025-04-04
- CAS:
- 434-90-2
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- 1Ton

US $101.00-1.00/KG2024-03-25
- CAS:
- 434-90-2
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- g-kg-tons, free sample is available