Application research of RU 58841
Introduction
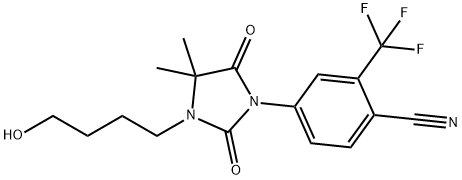
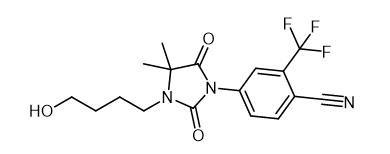
RU 58841(Figure 1) is a new non-steroidal antiandrogen which displays a high and specific binding to the androgen receptor, equivalent or higher than that of testosterone,in different animal species as well as in humans.Its affinity is about 30 times higher than that of other non-steroidal antiandrogens such as Anandron or flutamide. Its local antiandrogenic activity was evaluated on the Hamster F.O., which is widely used as a convenient screening model. Compared to the alternate rat sebaceous gland model, it has the advantage of allowing the measurement of the time-dependent decrease in F.O. area on the same animal. The F.O.s,consisting mainly of androgen-dependent sebaceous tissue, are strikingly decreased after castration. In vivo, when topically applied, it exerts a potent dose-dependent regression of F.O. area at a dose as low as 1 microgram/animal while being devoid of antiandrogenic activity on deep accessory sex organs and of any effect on testosterone level up to 100 micrograms/animal. In the same species, after subcutaneous administration, it induces at the dose of 300 micrograms/animal, a small decrease in F.O. area equivalent to that of 1 microgram applied topically and a weak systemic activity. In intact rats, no effects were observed up to 1 microgram/animal whatever the route of administration. These results suggest that RU 58841 might useful for the topical treatment of androgen-dependent skin disorders such as acne, androgenetic alopecia and hirsutism.[1]

Local inhibition of sebaceous gland growth by topically applied RU 58841
The biological activity of a series of nonsteroidal, pure androgen receptor inhibitors was compared using the Syrian hamster ear skin sebaceous gland model. RU 58841, RU 56187, RU 38882 and cyproterone acetate were applied topically for 4 weeks on the ventral ear pinna of sexually mature male Syrian hamsters. Their order of efficacy was as follows: RU 58841 > RU 56187 > RU 38882 > cyproterone acetate. Maximal reduction of 60% in the size of the sebaceous glands was observed in hamsters treated with RU 58841 at a dose of 10 micrograms per day. This degree of inhibition occurred without any systemic side effects as shown by the absence of inhibition on the contralateral untreated ear pinna. Longer treatment did not produce greater inhibition since extending the treatment period from 4 weeks to 12 weeks showed similar data. The effect of RU 58841 was reversible since the inhibited sebaceous glands returned to normal size within 4 weeks after the cessation of the topical applications. The potent localized inhibition of sebaceous glands by RU 58841 demonstrates the excellent potential of this compound as a topical drug for the treatment of acne and other androgen-mediated disorders.[2]
RU 58841-myristate--prodrug development
Acne and androgenetic alopecia are linked to androgen effects and therefore should improve following topical application of antiandrogens. We present a new antiandrogen prodrug, RU 58841-myristate (RUM) for topical therapy. Almost devoid of affinity to the androgen receptor, as derived from investigations in the mouse fibroblast cell line 29 +/GR +, RUM is rapidly metabolised to the potent antiandrogen RU 58841 by cultured human foreskin fibroblasts and keratinocytes, male occipital scalp skin dermal papilla cells, and by cells of the sebaceous gland cell line SZ95. In order to improve a specific targeting of the hair follicle, RUM was loaded on solid lipid nanoparticles (SLN), which are already known to support dermal targeting effects. Physically stable RUM loaded SLN were produced by hot homogenization. Penetration/permeation studies carried out using the Franz diffusion cell proved only negligible permeation of reconstructed epidermis and excised porcine skin within 6 h, implying a more topical action of the drug. Targeting to the hair follicle using SLN was visualised by fluorescence microscopy, following the application of Nile Red labelled SLN to human scalp skin. Transmission electron microscopy (TEM) allowed to detect intact silver labelled SLN in porcine hair follicles of preparations applied to the skin for 24 h. RUM loaded SLN should be considered for topical antiandrogen therapy of acne and androgenetic alopecia.[3]
The sebaceous gland pathway in the cutaneous permeation of RU 58841
The significance of the sebaceous gland pathway in the cutaneous permeation of an antiandrogen, 4-[3-(4-hydroxybutyl)-4,4-dimethyl -2,5-dioxo-1-imidazolidinyl]-2-(trifluoromethyl)benzonitrile (RU 58841), was studied with normal hairless rat skin and an induced scar hairless rat skin without sebaceous glands. RU 58841 was dissolved in an alcoholic solution and encapsulated in liposomes for comparison. After 24 h, the cumulative percentage of RU 58841 absorbed in vitro was 3-4-fold higher in the normal skin than in the scar skin; in the case of liposomes, the accumulation of the drug in the normal dermis was significantly higher than in the scar one. In the in vivo cutaneous distribution, the epidermis and dermis of the normal skin contained higher amounts of RU 58841 than the scar skin (ninefold with the solution and 16-fold with liposomes). An autoradiography study showed that with the solution, the drug was mainly localized in the stratum corneum/epidermis, and with the liposomes, the drug was mainly localized in the sebaceous glands. We concluded that the sebaceous glands constituted the main pathway for RU 58841. The alcoholic solution encouraged the localization of the drug into the stratum corneum, whereas liposomes targeted the sebaceous glands.[4]
The arylhydantoin RU 58841 derivatives synthesis
A series of side-chain derivatives of the arylhydantoin RU 58841 and the arylthiohydantoin RU 59063, wherein the aromatic trifluoromethyl group was replaced with iodine, was synthesized for possible development as radioiodinated androgen receptor (AR) ligands. Derivatives containing the cyanomethyl, methoxyethyl and propenyl side-chains displayed moderately high affinity (K(i)=20-59nM) towards the rat AR. Side-chains containing bulky lipophilic groups such as, benzyl and phenylpropyl, were poorly tolerated (K(i)>219nM). Superior AR binding affinities (0.71nM<K(i)<11nM) were displayed by arylhydantoins and arylthiohydantoin derivatives containing hydroxybutyl or methyl side-chains. The latter compounds are potential candidates for development as radioiodinated AR ligands.[5]
References
[1] Battmann T, Bonfils A, Branche C, et al. RU 58841, a new specific topical antiandrogen: a candidate of choice for the treatment of acne, androgenetic alopecia and hirsutism. J Steroid Biochem Mol Biol. 1994;48(1):55-60. doi:10.1016/0960-0760(94)90250-x
[2] Matias JR, Gaillard M. Local inhibition of sebaceous gland growth by topically applied RU 58841. Ann N Y Acad Sci. 1995;761:56-65. doi:10.1111/j.1749-6632.1995.tb31369.x
[3] Münster U, Nakamura C, Haberland A, et al. RU 58841-myristate--prodrug development for topical treatment of acne and androgenetic alopecia. Pharmazie. 2005;60(1):8-12.
[4] Bernard E, Dubois JL, Wepierre J. Importance of sebaceous glands in cutaneous penetration of an antiandrogen: target effect of liposomes. J Pharm Sci. 1997;86(5):573-578. doi:10.1021/js960394l
[5] Van Dort ME, Jung YW. Synthesis and structure-activity investigation of iodinated arylhydantoins and arylthiohydantoins for development as androgen receptor radioligands. Bioorg Med Chem Lett. 2004;14(21):5285-5288. doi:10.1016/j.bmcl.2004.08.031
You may like
Related articles And Qustion
Lastest Price from RU 58841 manufacturers

US $0.00/kg2026-03-27
- CAS:
- 154992-24-2
- Min. Order:
- 1kg
- Purity:
- 99.9
- Supply Ability:
- 10 tons

US $2000.00/kg2026-03-02
- CAS:
- 154992-24-2
- Min. Order:
- 1kg
- Purity:
- 99.9
- Supply Ability:
- 9999