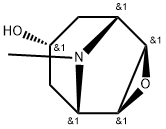
Application research of Scopine
Introduction
Scopine (Figure 1) is one of the metabolites of scopolamine which is a muscarinic antagonist used in the clinic to prevent motion sickness. No pharmacological activity of scopine was reported and our study established for the first time the role of scopine as a brain-targeting moiety. Scopine has a simple and well-defined structure as well as a low degree of toxicity compared with macromolecular based targeting moieties.[1]

Scopine enhances the brain uptake of chlorambucil
The blood brain barrier (BBB) represents the biggest challenge for therapeutic drugs to enter the brain. In this study, researchers selected chlorambucil (CHL), an alkylating agent, as the model therapeutic agent, and used scopine as a novel brain-targeting moiety. Herein, they synthesized Chlorambucil-Scopine (CHLS) prodrug and evaluated its brain-targeting efficacy. The tissue distribution study after i.v. injection revealed that the AUC0-t and Cmax of CHLS in the brain were 14.25- and 12.20-fold of CHL, respectively. Specifically, CHLS accumulated in bEnd.3 and C6 cells in an energy-dependent manner. In C6 cells, superior anti-glioma activity with a significantly decreased IC50 of 65.42 nM/mL was observed for CHLS compared to CHL (IC50 > 400 nM/mL). The safety evaluation, including acute toxicity, pathology, and hematology study, showed minimal toxicity toward nontargeting tissues, and also reached a lower systemic toxicity at 5 mg/kg (i.v.). these results suggested that scopine is a potential brain-targeting moiety for enhancing the brain uptake efficiency of CHL.[1]
Scopine Isolated in the Gas Phase
The rotational spectrum of the tropane alkaloid scopine is detected by Fourier transform microwave spectroscopy in a pulsed supersonic jet. A nonconventional method for bringing the molecules intact into the gas phase is used in which scopine syrup is mixed with glycine powder and the solid mixture is vaporized with an ultrafast UV laser beam. Laser vaporization prevents the easy isomerization to scopoline previously observed with conventional heating methods. A single conformer is unambiguously observed in the supersonic jet and corresponds to the energetically most stable species according to quantum chemical calculations. Rotational and centrifugal distortion constants are accurately determined. The spectrum shows fine and hyperfine structure due to the hindered rotation of the methyl group and the presence of a quadrupolar nucleus (14N), respectively. This additional information allows the angle of N-methyl inversion between the N-CH3 bond and the bicyclic C-N-C plane to be determined (131.8-137.8°), as well as the internal rotation barrier of the methyl group (6.235(1) kJ/mol ).[2]
The distorted tropane of scopoline
The structural isomerization of scopine into scopoline (oscine) has been observed in a supersonic jet expansion using microwave spectroscopy. Scopine has an epoxide group and this strained three-membered ring makesthe molecule more reactive. In fact scopine and its esters, in particular scopolamine (hyoscine, Scheme 1), are known to rearrange under alkaline conditions to scopoline (oscine), so additional model calculations were accomplished for the later isomer. These new predictions finally allowed the assignment of the experimental rotational transitions, confirming that scopoline was the carrier of the spectrum, and thus assessing the feasibility of the gas-phase intramolecular rearrangement reaction. Additional searches for other isomers or rotamers of scopoline were later verified, but researchers did not detect any additional species. The predicted spectra for the most stable species of both scopine and scopolineare compared with the experiment.
The rotational spectrum evidences a single structure in the gas phase, providing a first description of the (three-ring) structurally distorted tropane in scopoline. The absence of rotational signatures of any scopine conformation suggests a practically quantitative isomerization at the vaporization temperatures of the experiment (ca. 90 °C). The determined rotational parameters of scopoline reveal the structural consequences of the intramolecular cyclation of scopine, which breaks the original epoxy group and creates a new ether bridge and a 7β-hydroxytropane configuration. The hydroxy group further stabilizes the molecule by an O-H⋅⋅⋅N intramolecular hydrogen bond, which, in turn, forces the N-methyl group to the less stable axial form. Supporting ab initio (MP2) and DFT (B3LYP, M06-2X) calculations are included.[3]
References
[1] Wang X, Li J, Xu C, et al. Scopine as a novel brain-targeting moiety enhances the brain uptake of chlorambucil. Bioconjug Chem. 2014;25(11):2046-2054. doi:10.1021/bc5004108
[2] Écija P, Vallejo-López M, Uriarte I, et al. Scopine Isolated in the Gas Phase. Chemphyschem. 2016;17(19):3030-3034. doi:10.1002/cphc.201600368
[3] Écija P, Cocinero EJ, Lesarri A, Basterretxea FJ, Fernández JA, Castaño F. The distorted tropane of scopoline. Chemphyschem. 2013;14(9):1830-1835. doi:10.1002/cphc.201300199
You may like
See also
Lastest Price from Scopine manufacturers

US $0.00-0.00/KG2025-04-04
- CAS:
- 498-45-3
- Min. Order:
- 1KG
- Purity:
- 98%
- Supply Ability:
- 1ton
US $50.00/g2025-04-02
- CAS:
- 498-45-3
- Min. Order:
- 1g
- Purity:
- 97%
- Supply Ability:
- 50kg per month