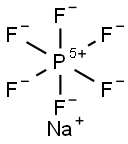Application research of sodium hexafluorophosphate
Introduction
Among the sodium battery electrolytes, sodium hexafluorophosphate (NaPF6) exhibits superior conductivity, anodic stability, and stable cathode electrolyte interface compared to other electrolytes. In sodium-ion batteries (SIBs), sodium hexafluorophosphate is dissolved in carbonate solvents and used as an electrolyte. The solubility of sodium hexafluorophosphate and suitability of a solvent mixture for sodium hexafluorophosphate are still debated. However, a detailed study confirmed that sodium hexafluorophosphate outperforms NaClO4 in SIBs and unlike the latter, the former is not an explosive. Most researchers prefer the solvent mixture,ethylenecarbonate:diethylcarbonate (EC:DEC), for dissolving sodium hexafluorophosphate for SIBs. The concentration of sodium hexafluorophosphate and subsequentformation of NaF, HF, and phosphates in the EC: DEC mixture is a major concern. These impurities formed due to hydrolysis of sodium hexafluorophosphate, have a deleterious effect on battery performance. Furthermore, the synthesis of sodium hexafluorophosphate itself is nontrivial. Usually,sodium hexafluorophosphate is synthesized using HF, which is cumbersome. For example,mixture of sodium and PCl5 was fluorinated with HF. In another method, PF5 was reacted with NaF to obtain sodium hexafluorophosphate. It must be noted that the PF5 was synthesized by reacting red phosphorous with HF and ClF3. Reaction of pyridinium hexafluorophosphate with sodium hydroxide produced sodium hexafluorophosphate.[1]
Mechanochemical Large-Scale Rapid Synthesis of Ultrapure Sodium Hexafluorophosphate
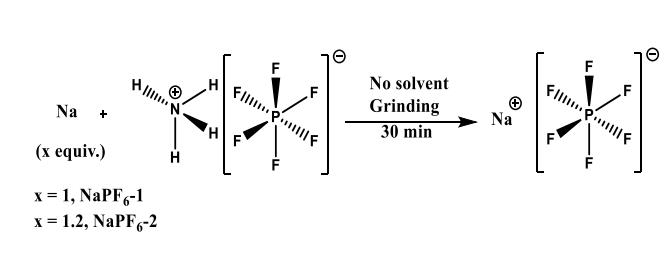
Usually, sodium hexafluorophosphate is synthesized using HF. The typical synthesis involves the grinding of one equivalent of sodium metal and ammonium hexafluorophosphate in a mortar and pestle (Scheme 1). During the reaction, hydrogen and ammonia evolve leading to the formation of sodium hexafluorophosphate. The grinding was carried out in an argon filled glove bag (Aldrich AtmosBag). In this approach, sodium hexafluorophosphate is synthesized by grinding dry ammonium hexafluorophosphate (NH4PF6) and sodium metal. Sodium injects an electron into the ammonium ion, which results in the formation of ammonia and hydrogen. The gram scale synthesis is completed in about 30 min. Purification of the product is not needed. The product purity is confirmed by various spectroscopic and electrochemical techniques. Usually, sodium hexafluorophosphate comprises NaF,HF, and solvents as impurities that affect the performance of SIBs. It has been confirmed that the sodium hexafluorophosphate synthesized by their mechanochemical approach in the absence of solvent is devoid of impurities despite the absence of product purification step.Furthermore, the synthesis of pure sodium hexafluorophosphate (250 g) is demonstrated using a grinder used as household item in cooking Indian pancakes, which costs about 300 USD. The duration of the synthesis of 250 g pure sodium hexafluorophosphate is 1 h. The purity of this sample is comparable to that of sodium hexafluorophosphate (5 g) synthesized using mortar and pestle.[1]
Sodium hexafluorophosphate mediated enhancement of electrochemical properties of poly(vinyl alcohol)–chitosan solid polymer electrolytes
Sodium salts, such as sodium hexafluorophosphate, are integral to the development of solid polymer electrolytes for sodium-ion batteries. When combined with polymers like PEO, PVA and PVDF–HFP, sodium hexafluorophosphate enhances ionic conductivity. For instance, the PVA–sodium hexafluorophosphate (with PVA : sodium hexafluorophosphate weight ratio of 60 : 40) exhibits the highest room-temperature ionic conductivity, reaching 3.65 ×10−5 S cm−1. sodium hexafluorophosphate is the preferred electrolyte salt because of its high ionic conductivity, electrochemical stability, and compatibility with electrode materials. Research indicates sodium hexafluorophosphate-based electrolytes achieve ionic conductivities of 5–7mS/cm at room temperature and remain stable up to 4.5 V vs.Na/Na+. Additionally, sodium hexafluorophosphate facilitates favorable surface film formation and enhances cathode material reversibility. Compared with NaClO4, sodium hexafluorophosphate offers superior Na+ desolvation kinetics and better interfacial stability. These properties make sodium hexafluorophosphate a strong candidate for sodium-ion battery electrolytes,combining high performance, stability, and conductivity. sodium hexafluorophosphate-based polymer electrolytes also enhance the performance of electric double-layer capacitors (EDLCs) by expanding their voltage window beyond 3.5 V, leading to improved energy density.
In this study, a free-standing, flexible and biodegradable biopolymer electrolyte (BPE) derived from a poly(vinyl alcohol) (PVA)-chitosan (CS) blend immobilizing sodium hexafluorophosphate (NaPF6) salt was fabricated via solution casting method. The effect of salt concentration on the structural, electrical, and electrochemical properties of the electrolyte was systematically investigated. X-ray diffraction (XRD) and Fourier-transform infrared (FTIR) spectroscopy were used to ascertain the microstructural changes in the polymer matrix including the complexation of PVA, CS, and NaPF6. Electrochemical impedance spectroscopy (EIS) measurements revealed that the BPE containing 40 wt% NaPF6 exhibited the highest conductivity (6.94 ± 0.04) × 10-5 S/cm, which was three-order enhancement over the pristine system. The ion transport behaviour, interpreted through the Schütt and Gerdes (S-G) model, revealed that the ionic conductivity of the SPE system is strongly influenced by both the concentration of charge carriers and their mobility. The electrolyte displayed a predominant ionic nature with an electrochemical stability window of ∼3.25 V. When incorporated into an Na-ion EDLC, the optimized electrolyte sample provided a specific capacitance of 42.65 F/g, energy density of 5.4 W h/kg, and power density of 95 W kg-1, as determined by galvanostatic charge-discharge (GCD) tests performed at 0.05 mA/g.[2]
A Highly Reversible Room-Temperature Sodium Metal Anode
Owing to its low cost and high natural abundance, sodium metal is among the most promising anode materials for energy storage technologies beyond lithium ion batteries. However, room-temperature sodium metal anodes suffer from poor reversibility during long-term plating and stripping, mainly due to formation of nonuniform solid electrolyte interphase as well as dendritic growth of sodium metal. Herein researchers report for the first time that a simple liquid electrolyte, sodium hexafluorophosphate in glymes (mono-, di-, and tetraglyme), can enable highly reversible and nondendritic plating-stripping of sodium metal anodes at room temperature. High average Coulombic efficiencies of 99.9% were achieved over 300 plating-stripping cycles at 0.5 mA/cm2. The long-term reversibility was found to arise from the formation of a uniform, inorganic solid electrolyte interphase made of sodium oxide and sodium fluoride, which is highly impermeable to electrolyte solvent and conducive to nondendritic growth. As a proof of concept, researchers also demonstrate a room-temperature sodium-sulfur battery using this class of electrolytes, paving the way for the development of next-generation, sodium-based energy storage technologies.
References
[1] Senthilkumaran M, Javaregowda BH, Babu Rajendran P, et al. Mechanochemical Large-Scale Rapid Synthesis of Ultrapure Sodium Hexafluorophosphate. Chempluschem. 2025;90(7):e202400640. doi:10.1002/cplu.202400640
[2] Cyriac V, Ismayil, Mishra K, et al. Sodium hexafluorophosphate mediated enhancement of electrical and electrochemical properties of poly(vinyl alcohol)-chitosan solid polymer electrolytes for EDLCs. RSC Adv. 2025;15(30):24350-24366. Published 2025 Jul 11. doi:10.1039/d5ra02897c
[3] Seh ZW, Sun J, Sun Y, Cui Y. A Highly Reversible Room-Temperature Sodium Metal Anode. ACS Cent Sci. 2015;1(8):449-455. doi:10.1021/acscentsci.5b00328
You may like
Lastest Price from Sodium hexafluorophosphate manufacturers

US $5.00/kg2025-04-21
- CAS:
- 21324-39-0
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 10000

US $9.90/KG2025-04-21
- CAS:
- 21324-39-0
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt