Application research of Ziconotide acetate
Introduction
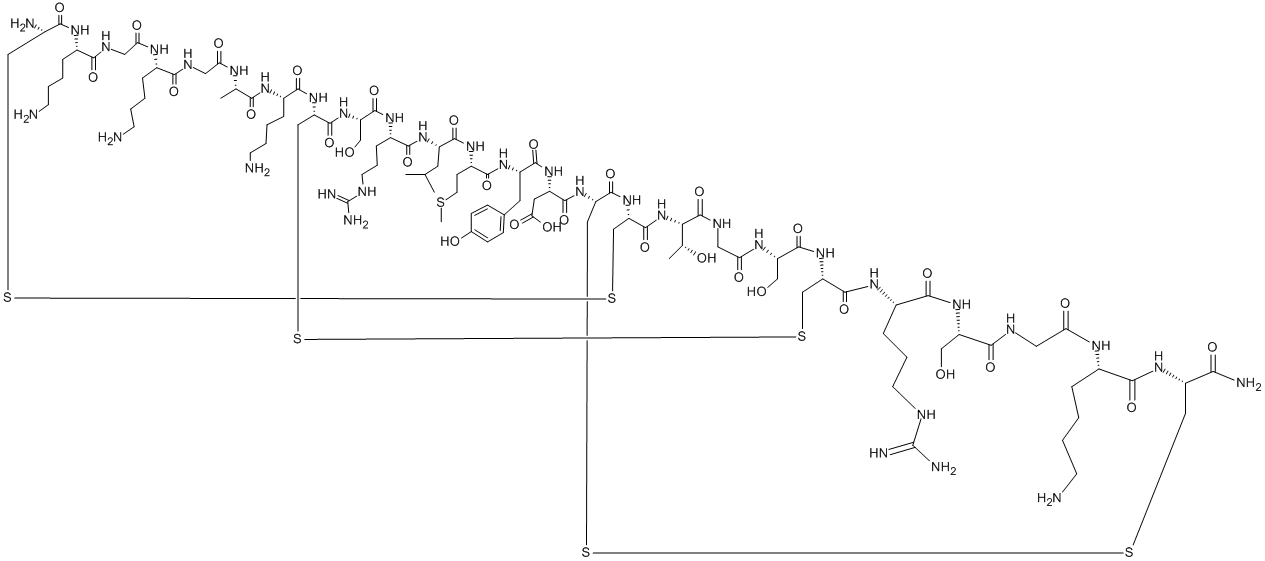
Ziconotide acetate is a synthetic version of the hydrophilic conopeptide ω-conotoxin MVIIA, originally isolated from the marine snail Conus magus in 1982 while it inhabited the Pacific Ocean]. The slow-paced Conus magus employs specific hunting strategies to capture fast-moving fish, slower-moving mollusks, and worms by shooting hollow radular teeth filled with poisonous peptides. It is the inaugural member of a new drug class known for its potent inhibitory activity on N-type voltage-gated calcium channels, with minimal affinity for other ion channels and no interaction with cholinergic, monoaminergic,or peptidergic receptors. In June 2000, Elan received approval from the US Food and Drug Administration (FDA) for the intrathecal injection of ziconotide acetate as a pain medication, with the Phase III ischemia trial still ongoing. In 2004, the FDA officially licensed ziconotide acetate for intrathecal administration in the treatment of severe pain. Ziconotide acetate has a molecular formula of C102H172N36O32S7, a relative molecular weight of 2639.12, a CAS chemical identifier number of 107452-89-1, and an isoelectric point of 11.2. The molecule consists of 25 highly hydrophilic amino acids arranged in the following sequence: H-Cys-Lys-Gly-Lys-Gly-Ala-Lys-Cys-Ser-Arg-Leu-Met-Tyr-Asp-Cys-Cys-Thr-Gly-Ser-Cys-Arg-Ser-Gly-Lys-Cys-NH2 cyclic (1–16), (8–20), (15–25)-tris(disulfide). These amino play a crucial role in stability of molecule and includes essential amino acids, including four Lysine(Lys) and two Arginine (Arg) residues. Additionally, the molecule exists as a cation in bodily fluids. In non-in vivo settings, free L-methionine, which exists in the vials (50ug/ml), is used as the vehicle for ziconotide because it is more easily oxidized compared with the methionine in ziconotide.[1]
Mechanism of action of ziconotide acetate
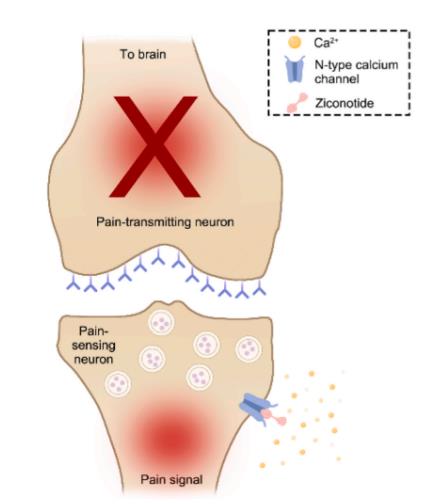
Unlike opioids, which primarily act by binding to specific receptors or regulating receptor levels associated with pain, ziconotide acetate alleviates pain by modulating synaptic function. Specifically, ziconotide acetate inhibits N-type calcium channels in the spinal cord, leading to reduced release of pain-relevant neurotransmitters in the central terminals of primary afferent neurons(Fig. 1). Due to its unique structure and acting sites, ziconotide cannot penetrate the Blood-Brain Barrier (BBB),necessitating intrathecal administration as the approved route of delivery.Ziconotide acetate binds to N-type calcium channels located on the primary nociceptive afferent neurons in the superficial layers of the dorsal horn in the spinal cord. Ziconotide binding blocks calcium entry into the presynaptic nerve terminal, thereby reducing the release of excitatory neurotransmitters from the primary afferent nerves terminals into the synapse. Physically, ziconotide acetate lodges into the permeation pathway of the channels as a pore blocker with a quite low unblocking rate. [1]
Ziconotide acetate and psychosis
Ziconotide acetate is a non-opioid analgesic that acts on N-type voltage-gated calcium channels. Despite its proven effectiveness in pain treatment, it can induce neuropsychiatric symptoms. The aim of this article is to present a case of psychosis secondary to ziconotide acetate and to explore the variety of neuropsychiatric symptoms it produces, exploring the relationship between these symptoms and the mechanism of action of ziconotide acetate. For this purpose, a clinical case is presented as well as a scoping review of other cases published in the scientific literature. A search on Web of Science, Pubmed and Embase databases was performed on December 11, 2023, following the criteria of the PRISMA-ScR Statement. The clinical case presented shows the variety of neuropsychiatric symptomatology that ziconotide acetate can cause in the same patient. On the other hand, 13 papers were retrieved from the scoping review (9 case reports, 4 case series), which included 21 cases of patients treated with ziconotide acetate who presented adverse effects ranging from psychotic symptoms to delirium. In conclusion, the variety of neuropsychiatric symptoms derived from ziconotide acetate could be related to the blockade of N-type voltage-gated calcium channels in glutamatergic and GABAergic neurons, in turn affecting dopaminergic pathways.[2]
Intrathecal ziconotide acetate for the treatment of chronic pain
Despite the wide use of ziconotide acetate in the USA for treating refractory cancer- and noncancer-related pain, this agent is little used in Europe, even if licensed by the European Medicines Agency (EMA). The reason could be attributed to the high, fixed starting dose required for ziconotide acetate, as stated in the EMA Summary of Product Characteristics (SmPC). This dosage recommendation is based on the results of pivotal clinical studies of ziconotide acetate, which utilized aggressive titration schedules. Thus, a reappraisal of the available evidence, as well as a reflection on real-life clinical experiences, might be useful to identify practice adjustments to improve the clinical application of ziconotide acetate in the European scenario. In line with this need, this paper reports some clinical experiences of patients with chronic pain treated with ziconotide acetate intrathecal (IT) therapy in Italy, particularly focusing on long-term treatment to further characterize and improve the use of this agent in real practice. Moreover, a literature review of the available data on the effectiveness and safety of IT ziconotide acetate is provided. Collected clinical experiences suggested that the use of IT ziconotide acetate represents a valuable option, particularly in cases where other treatments have been ineffective or poorly tolerated. Ziconotide acetate was shown to not cause severe side effects in the long-term treatment, leading to a constant pain relief effect at stable doses, without adverse events that caused therapy interruption. The overall constant ziconotide acetate dosages also suggest the absence of a tolerance effect. In parallel, the evidence in the literature aligns with real-world evidence and further supports the use of IT ziconotide acetate as an important option for the management of chronic pain.
IT ziconotide acetate represents a valuable addition to the armamentarium of pain management strategies, offering hope for improved quality of life for patients suffering from chronic, treatment-resistant pain. Continued research and clinical experience will further elucidate its optimal use and role in comprehensive pain care.
References
[1] Lin J, Chen S, Butt UD, Yan M, Wu B. A comprehensive review on ziconotide. Heliyon. 2024;10(10):e31105. Published 2024 May 11. doi:10.1016/j.heliyon.2024.e31105
[2] Peraire M, Gimeno-Vergara R, Pick-Martin J, Boscá M, Echeverria I. Ziconotide and psychosis: from a case report to a scoping review. Front Mol Neurosci. 2024;17:1412855. Published 2024 Oct 16. doi:10.3389/fnmol.2024.1412855
[3] De Negri P, Mastronicola C. Intrathecal ziconotide for the treatment of chronic pain: a collection of clinical experiences and literature review. Eur Rev Med Pharmacol Sci. 2024;28(15):4105-4110. doi:10.26355/eurrev_202408_36640
You may like
Lastest Price from Ziconotide Polyacetate manufacturers

US $1000.00-1000.00/g2025-09-19
- CAS:
- 107452-89-1
- Min. Order:
- 1g
- Purity:
- >=99% (HPLC), USP43
- Supply Ability:
- 10000000g

US $0.00/mg2025-04-21
- CAS:
- 107452-89-1
- Min. Order:
- 1mg
- Purity:
- 99%
- Supply Ability:
- 10kg/month