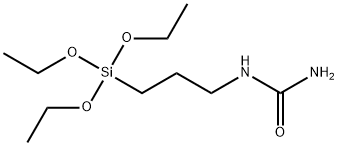
Applications of N-(Triethoxysilylpropyl)urea
N-(Triethoxysilylpropyl)urea is a class of organosilicon compounds featuring both urea and siloxane functional groups. At ambient temperatures, this substance exists as a colourless to pale yellow transparent liquid, readily soluble in organic solvents such as ethanol and diethyl ether. Upon contact with water, it undergoes gradual hydrolysis to form silanol groups whilst releasing ethanol. As a functional silane coupling agent, its primary function is to enhance interfacial compatibility between inorganic and organic materials. Widely employed in coatings, adhesives, and composite materials, N-(Triethoxysilylpropyl)urea significantly improves adhesion, water resistance, and mechanical strength. It is also utilised in textile modification to impart antibacterial and antistatic properties to substrates.

Eco-friendly flame-retardant coatings based on N-(Triethoxysilylpropyl)urea
Cotton fabrics have been used widely with the advantages of softness, comfort, etc. However, cotton fabrics are extremely flammable. Once ignited, the flames can spread rapidly and are difficult to extinguish. In recent years, the frequency of cotton fabrics-related fires has increased, posing a serious threat to human life and resulting in significant property damage. Hence, the improvement of cotton fabrics' flame retardancy has drawn more concern and study from both industry and academics. The above facts indicated that the performance of PA and APTES in enhancing the cotton fabrics' flame retardancy was outstanding, while PA simultaneously decreased the mechanical properties of cotton fabrics. This decrease can be attributed to PA's acidity. In recent years, researchers have developed N-(Triethoxysilylpropyl)urea (TESPR) based on APTES to improve the adhesive properties and mechanical properties of materials, which is used as an adhesion enhancer, a surface modifier, a cross-linking binder and a dispersant. It is mainly used to improve the adhesion and compatibility of fiber-to-polymer (resin) adhesion, the adhesion and water resistance of resin coatings to an inorganic substrate. However, there have been limited reports on the flame retardancy of cotton fabrics treated with N-(Triethoxysilylpropyl)urea and PA using the sol-gel technique. Hence, with the sol-gel technique, the cotton fabrics' flame retardancy was enhanced in this study constructed by N-(Triethoxysilylpropyl)urea and AP. The analyses of surface morphology, flame retardant properties, thermal degradation properties and mechanical properties of samples were explored and yielded some noteworthy results.[1]
The breaking force of Cotton/TESPR/AP was dramatically enhanced compared with that of Cotton/AP, with the presence of N-(Triethoxysilylpropyl)urea. The breaking force in warp of Cotton/TESPR/AP was measured to be 299 N, while the breaking force in weft was 282 N. Uniform deposition of TESPR/AP was verified by SEM combined with EDX for coated cotton fabrics. The char residues of Cotton/ AP and Cotton/TESPR/AP were more than that of Cotton at 700 °C, illustrating that TESPR/AP promoted the carbonization of cotton fabrics. Deposition of N-(Triethoxysilylpropyl)urea and AP significantly enhanced the cotton fabrics' flame retardancy. Cotton/TESPR/AP exhibited an immediate self-extinguishing effect upon removal of the igniter with LOI value of 31.0%. Additionally, the TESPR/AP coatings contributed to the reduction of PHRR, THR, TSP and PSPR of coated cotton fabrics. The analysis of TG-FTIR revealed the release of a higher amount of nonflammable gases, including H2O and CO2, while fewer flammable gases were detected for Cotton/TESPR/AP. These findings indicated a notable improvement in the flame retardancy of Cotton/TESPR/AP. Breaking force results showed that compared with that of Cotton/AP, the breaking force of Cotton/TESPR/AP had been significantly improved. The results of handle feel and air permeability results indicated that Cotton/TESPR/AP still retained good air permeability and whiteness, as well as drape, wrinkle recover, softness and resilience whites. This study showed that Cotton/ N-(Triethoxysilylpropyl)urea/AP had great potential for ecological flame retardancy of cotton fabrics and versatile finishing applications.
Functionalization of periodic mesoporous organosilica
As a novel class of organic–inorganic nanocomposites, periodic mesoporous organosilicas have drawn much attention due to potential applications such as host-guest inclusion, nanotechnology, chemoselective separation and adsorption, chemical sensing, and catalysis. These PMOs materials are synthesized by the supramolecular–assembly route in the presence of structure-directing agents using a silsesquioxane of the type (EtO)3Si–R–Si(OEt)3 as the sole precursor, therefore the organic groups R are located within the channel walls as bridges between Si centers. Functionalized mesoporous materials were prepared using BTESE as the source of bridging organic groups, N-(Triethoxysilylpropyl)urea as the source of terminal organic groups, and P123 as the structure-directing agent. For a typical synthesis, a molar ratio of BTESE, 1 minus x; N-(Triethoxysilylpropyl)urea x; P123, 0.034; HCl, 11.7; H2O, 326 was used, where x varies from 0% to 40%. In a typical procedure, a solution of P123, HCl and water was prepared at 40 °C. To this solution was added dropwise a mixture of BTESE and N-(Triethoxysilylpropyl)urea. The mixture was allowed to stir for 24 h until white precipitates formed, and then the products were moved into Teflon-lined autoclaves and aged for 72 h at 100 °C. The white precipitates were recovered by filtration, washed with water until reaching a pH of 6–7, and then dried at atmosphere. The surfactant template was removed from organosilica materials through solvent extraction. An as-synthesized sample (0.5 g) was gently stirred for 6 h in a solution of HCl and ethanol in 50 °C water bath. This procedure was repeated several times until the surfactants were totally removed. The powder was filtered, washed with ethanol, and air-dried at 60 °C overnight to obtain final material.[2]
Functionalized materials have an excellent mesoscopic order as the concentration of N-(Triethoxysilylpropyl)urea is equal to 20 mol%, which seems to be incompatible to the above-mentioned mechanism. Such surprising observation may be related to the special nature of ureidopropyl groups in solution. As the concentration of UPTES increases gradually, a fraction of UPTES tends to form the so-called oligomerized silicate species through the intermolecular condensation of the hydrolysate of N-(Triethoxysilylpropyl)urea. The oligomerized silicate species, consisting of hydrophobic moieties and hydrophilic sections, have a similar behavior in solution as the organosilicon surfactants. On the basis of the co-condensation of bis(triethoxysilyl)ethane and ureidopropyltriethoxysilane, ureidopropyl groups are terminally functionalized to periodic mesoporous organosilicas without any damage to the ethane bridging groups in the framework. The functionalized materials become structurally disordered with increasing of ureidopropyl group except for the sample with 20 mol% N-(Triethoxysilylpropyl)urea, which displays a highly ordered two-dimensional hexagonal structure, with a better mesoscopic order than that of pure PMOs. This material still possesses a desirable pore structure with a surface area of 565 m2/g, a pore volume of 1.1 cm3/g, and a mean pore size of 10.1 nm, although it shows a trend that the surface area and pore volume decrease with increasing amount of UPTES for functionalized materials.
References
[1]Liu, H., Li, P., Xu, Y.-J., Zhu, P., & Liu, Y. (2024). Eco-friendly flame-retardant coatings based on γ-ureidopropyltriethoxysilane for cotton fabrics with improved flame retardancy and mechanical properties. Sustainable Materials and Technologies, 39, e00821.
[2]Wei, Q., Liu, L., Nie, Z.-R., Chen, H.-Q., Wang, Y.-L., Li, Q.-Y., & Zou, J.-X. (2007). Functionalization of periodic mesoporous organosilica with ureidopropyl groups by a direct synthesis method. Microporous and Mesoporous Materials, 101(3), 381–387.
Lastest Price from N-(Triethoxysilylpropyl)urea manufacturers

US $0.00-0.00/KG2025-06-17
- CAS:
- 23779-32-0
- Min. Order:
- 0.01KG
- Purity:
- 99.99%
- Supply Ability:
- 200T

US $0.00-0.00/KG2025-04-21
- CAS:
- 23779-32-0
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt