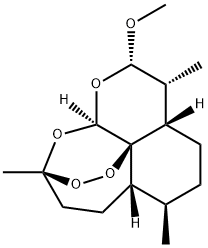
Artemether: Antimalarial Efficacy, Pharmacokinetics & Toxicity
Artemether is an artemisinin derivative that is artemisinin in which the lactone has been converted to the corresponding lactol methyl ether. It is used in combination with lumefantrine as an antimalarial for the treatment of multi-drug resistant strains of falciparum malaria. This combination therapy exerts its effects against the erythrocytic stages of Plasmodium spp. and may be used to treat infections caused by P. falciparum and unidentified Plasmodium species, including infections acquired in chloroquine-resistant areas. Artemether has a role as an antimalarial. It is a sesquiterpenoid, a cyclic acetal, an organic peroxide, an artemisinin derivative and a semisynthetic derivative.

Artemether for severe malaria
Malaria is a febrile illness caused by Plasmodium parasites, which are transmitted to humans through the bite of infected female anopheline mosquitoes. Five species of Plasmodium cause this disease in humans, of which P. falciparum is the most common worldwide, and is responsible for almost all of the severe disease and deaths. Unlike artesunate, artemether is poorly soluble in water and the parenteral formulation is only available as a pre‐mixed oil‐based solution for intramuscular injection (80 mg/mL for use in adults and 40 mg/mL for children). The standard dose is 3.2 mg/kg on admission followed by 1.6 mg/kg once daily until oral therapy is tolerated (WHO 2010). Peak plasma concentrations typically occur around six hours after intramuscular injection, but in severely ill children with poor peripheral perfusion, absorption can be highly erratic. Conversely, artesunate is supplied as a dry powder for mixing with sodium bicarbonate prior to either intravenous or intramuscular injection (WHO 2010). Compared with artemether, the absorption of artesunate is more reliable with peak plasma concentrations following intramuscular injection occurring at around one hour (Hien 2004; Illet 2002; Nealon 2002). These more favourable pharmacokinetic properties of artesunate moved research attention away from artemether and artesunate now has a stronger evidence‐base and is the preferred therapy.[1]
We included 18 RCTs, enrolling 2662 children and adults with severe malaria. Eleven trials were conducted in Africa and seven trials were undertaken in Asia. For children (trials mostly conducted in Africa), there is probably little or no difference in the risk of death between intramuscular artemether and quinine (moderate quality evidence). For older children (> 15 years) and adults in Asia, it probably reduces deaths compared with quinine (moderate quality evidence), but larger trials are required to have full confidence in this finding. The two trials directly comparing artemether and artesunate were conducted in adults in Asia, and the results are therefore poorly applicable to children in Africa. However, in the absence of direct comparisons in children, the low quality evidence of equivalence between it and quinine suggests that artesunate will be as superior to it as it has been shown to be superior to quinine. This medicine is prone to erratic and partial absorption and takes longer to achieve peak plasma concentrations as demonstrated in animal and human studies. These pharmacokinetic attributes make it that is injected intramuscularly less readily available in the human body and may explain the difference in outcomes between artesunate and intramuscular artemether we have observed in this review.
Artemether Bioavailability after Oral or Intramuscular Administration
Oral artemether is absorbed rapidly and reliably. The antimalarial activity-time profile after oral administration of it was similar to that described previously, as determined by the same bioassay technique. Maximum antimalarial concentrations occurred within 3 h of administration. During convalescence the significantly lower AUC0-∞ of oral artemether and DHA combined probably resulted from expansion of the apparent volume of distribution and improved systemic clearance on recovery with increased presystemic (first-pass) intestinal and hepatic metabolism. This study cannot distinguish confidently between the pharmacokinetic effects of disease-reducing clearance in the acute phase or autoinduction-increasing clearance in the convalescent phase, although only three doses were given over >7 days, so the latter explanation seems less likely. Oral administration during the acute phase gave peak antimalarial activities which were approximately 16 times higher than those after intramuscular administration. Intramuscular artemether dosing was also associated with a considerable delay to the Cmax. The relative bioavailability of antimalarial activity during the first 24 h, the period that is likely to be the most important clinically, of oral medicine given during the acute phase was over three times that of intramuscular medicine.[2]
The one patient with no detectable antimalarial activity after the administration of intramuscular artemether may have been a nonabsorber, although an error in drug administration cannot be entirely excluded. The reduced antimalarial activity after intramuscular administration reflects slow absorption from the oil depot at the injection site. The approximately threefold reduction in the AUC following intramuscular administration is explained by the lower level of biotransformation of it to the metabolite DHA, which is approximately three times more potent than the parent drug as an antimalarial in vitro. As intramuscular artemether is absorbed so slowly, the 24-h sampling time may be insufficient to characterize the profile of the concentrations in plasma and therefore may underestimate absorption and, thus, total AUC. However, from a therapeutic standpoint it is the antimalarial activity in blood in the hours following the first administration of a parenteral drug that is critical, particularly in patients with severe malaria. The rate of absorption of the injected drug from the oil depot may be influenced by the oil itself. Use of alternative oils to dissolve these lipophilic drugs may accelerate absorption. On clinical and pharmacokinetic grounds, it is likely that the more potent intramuscular or intravenous artesunate would be superior to intramuscular artemether for the parenteral treatment of falciparum malaria.
Mechanisms of artemether toxicity on single cardiomyocytes
Malaria is a mosquito‐borne infectious disease caused by protozoa of genus Plasmodium sp., which represents a major health problem. According to the World Health Organization (WHO), malaria is endemic in nearly 100 countries and territories, particularly in underprivileged areas of Africa, Asia, and Latin America. In 2018, there were 228 million cases of malaria and an estimated 405,000 malaria deaths (WHO, 2018). In absence of an effective malaria vaccine, chemotherapy is still an unavoidable strategy to fight the disease. Unfortunately, the resistance of Plasmodium falciparum to several antimalarial drugs, such as chloroquine and mefloquine, is increasing dramatically. NCs containing artemether (NC‐ATM) were effective in combating malaria, achieving both cure and increasing the survival rate of mice by oral route. Given that, it was necessary to understand the effect of free artemether on cardiomyocytes. We focused on the excitation–contraction coupling of single cardiomyocytes from mice, and we investigated the mechanisms involved.[3]
Here, we report critical cellular effects of repeated oral administration of artemether for 4 days in mice. The in vivo treatment reproduced the effects of acute myocyte exposure to free artemether on AP duration and abnormal Ca2+ events. Overall, these results are in line with a direct and rapid effect of the drug. This also excluded the possibility that the effects seen after repeated administration in vivo result from cardiac remodelling, unlikely to occur within the 4‐day treatment, which was confirmed by our investigation of heart morphology and function by echocardiography. Incidentally, the reduction of myocyte contraction after treatment with free artemether was not found on myocardial function, either because this effect is modest or because the negative inotropy is compensated by one or more mechanisms external to the cell, such as neurohormonal system(s).
References
[1]Esu E, Effa EE, Opie ON, Uwaoma A, Meremikwu MM. Artemether for severe malaria. Cochrane Database Syst Rev. 2014 Sep 11;2014(9):CD010678. doi: 10.1002/14651858.CD010678.pub2. Update in: Cochrane Database Syst Rev. 2019 Jun 18;6:CD010678. doi: 10.1002/14651858.CD010678.pub3. PMID: 25209020; PMCID: PMC4455227.
[2]Silamut K, Newton PN, Teja-Isavadharm P, Suputtamongkol Y, Siriyanonda D, Rasameesoraj M, Pukrittayakamee S, White NJ. Artemether bioavailability after oral or intramuscular administration in uncomplicated falciparum malaria. Antimicrob Agents Chemother. 2003 Dec;47(12):3795-8. doi: 10.1128/AAC.47.12.3795-3798.2003. PMID: 14638485; PMCID: PMC296187.
[3]Moreira Souza AC, Grabe-Guimarães A, Cruz JDS, Santos-Miranda A, Farah C, Teixeira Oliveira L, Lucas A, Aimond F, Sicard P, Mosqueira VCF, Richard S. Mechanisms of artemether toxicity on single cardiomyocytes and protective effect of nanoencapsulation. Br J Pharmacol. 2020 Oct;177(19):4448-4463. doi: 10.1111/bph.15186. Epub 2020 Aug 24. PMID: 32608017; PMCID: PMC7484510.
You may like
Lastest Price from Artemether manufacturers

US $2500.00/Kg2025-11-09
- CAS:
- 71963-77-4
- Min. Order:
- 0.100Kg
- Purity:
- 99 %
- Supply Ability:
- 5000 Kg

US $0.00/kg2025-04-27
- CAS:
- 71963-77-4
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 1000kg