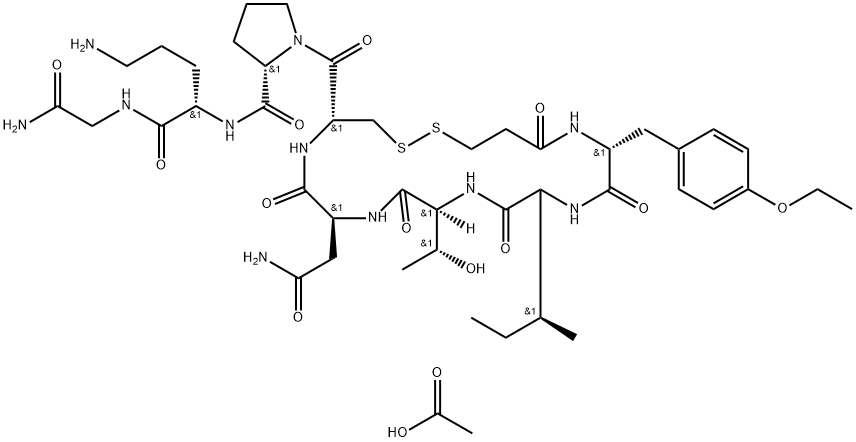
Atosiban Acetate: Tocolytic Mechanism and Clinical Evaluation
Atosiban Acetate is an inhibitor of the hormones oxytocin and vasopressin. It is used intravenously to halt premature labor. Although initial studies suggested it could be used as a nasal spray and hence would not require hospital admission, it is not used in that form. Atobisan was developed by the Swedish company Ferring Pharmaceuticals. It was first reported in the literature in 1985. Atosiban Acetate is licensed in proprietary and generic forms for the delay of imminent pre-term birth in pregnant adult women. It binds to membrane bound oxytocin receptors on the myometrium and prevents oxytocin-stimulated increases in inositol triphosphate production. This ultimately prevents release of stored calcium from the sarcoplasmic reticulum and subsequent opening of voltage gated calcium channels. This shutdown of cytosolic calcium increase prevents contractions of the uterine muscle, reducing the frequency of contractions and inducing uterine quiescence.

The effectiveness and safety of treatment of atosiban acetate
With the continuous advancement of assisted reproductive technology, the global incidence of twin pregnancies has significantly increased. In the United States, the rate of twin pregnancies in 2021 was reported to be 3.15%. Atosiban Acetate, a dual oxytocin/vasopressin V1A antagonist approved for clinical use in women at risk of imminent preterm labor, is an alternative option. It works by competing with oxytocin for receptor binding in the myometrium, decidua, and fetal membranes, thereby reducing oxytocin’s efficacy and calcium ion levels in muscle cells, which helps to inhibit uterine contractions, and there are no absolute contraindications to its use. There is limited research available on the use of atosiban in multiple pregnancies and the recommendations for preterm labor management. The efficiency and safety of continuous and repeated treatment with Atosiban Acetate in twin pregnancies remain unclear. Therefore, the aim of this study was to assess the effects and maternal safety of continuous and repeated treatment with atosiban in twin pregnancies, while monitoring maternal and neonatal morbidities. The findings of this study may provide valuable insights for clinical treatment strategies.[1]
The aim of this study was to assess the effect and adverse outcomes of continuous and repeated treatment with Atosiban Acetate in twin pregnancies. The main findings were as follows: (1) Women who received continuous and repeated treatment with atosiban experienced significant prolongation of pregnancy; (2) Neonates born after continuous and repeated treatment with atosiban had higher Apgar scores at birth, and the rate of NICU admission was reduced; and (3) There were no major antenatal or postnatal adverse outcomes for women who received continuous and repeated treatment. In this study, the mean duration of medication was 189.24 hours (equivalent to nearly 4 treatments of atosiban), and it effectively prolonged the latency period by 411.45 hours and the gestational age at birth by 1.5 weeks. Atosiban Acetate, an oxytocin receptor antagonist, was developed to delay preterm birth. In normal parturition, oxytocin stimulates contractions by converting phosphatidylinositol to inositol triphosphate, which binds to a protein in the sarcoplasmic reticulum, causing the release of calcium into the cytoplasm. Oxytocin receptor antagonists compete with oxytocin for binding to oxytocin receptors in the myometrium and decidua, thereby preventing the increase in intracellular free calcium that occurs when oxytocin binds to its receptor. Continuous and repeated treatment with atosiban demonstrated significant efficacy in prolonging pregnancy for twin pregnancies at risk of preterm labor. Continuous and repeated treatment with atosiban acetate was associated with lower fetal distress and lower NICU admission but no effect in adverse neonatal outcomes as well as the incidence of chorioamnionitis.
Atosiban Acetate versus placebo in the treatment of threatened preterm birth
In 2018, the WHO reported that preterm birth is the leading cause of death worldwide for children under the age of 5 years. Although various pharmacological agents and therapeutic interventions have been developed to reduce occurrence of preterm birth, preterm birth rates remain high. Various tocolytic drugs have shown to be effective in delaying delivery, including Atosiban Acetate, however, no tocolytic drug has proven to be effective in reducing neonatal morbidity and mortality compared with a placebo. Therefore, the current use of tocolytic drugs in case of threatened preterm birth is insufficiently supported by available evidence. For this reason, our research group started the double-blinded randomised controlled Assessment of Perinatal Outcome after Specific Tocolysis in Early Labour (APOSTEL) 8 trial in 2017. This trial evaluates the effectiveness of atosiban versus placebo administered for 48 hours in women with a diagnosis of threatened preterm birth between 30 and 34 weeks of gestation on neonatal morbidity and perinatal mortality. This follow-up study aims to determine the long-term effects of atosiban versus placebo in threatened preterm birth on neurodevelopment, behaviour problems and general health of children around 4 years of corrected age. The APOSTEL 8 follow-up will help to identify potential beneficial or harmful effects of Atosiban Acetate administration during pregnancy and will contribute to further improving threatened preterm birth healthcare.[2]
Eligible women are randomised to atosiban acetate or placebo (0.9% saline) for 48 hours. The primary outcome is a composite of adverse perinatal outcomes including perinatal mortality and six severe perinatal morbidities: bronchopulmonary, periventricular leukomalacia >grade 1, intraventricular haemorrhage >grade 2, necrotising enterocolitis ≥stage 2, retinopathy of prematurity >grade 2 or needing laser therapy, andculture-proven sepsis. Secondary outcomes include various infant and maternal outcomes which can be found in the published protocol. The APOSTEL 8 started in 2017 and complete recruitment in 2023. This long-term follow-up study is directed at the children born to mothers who participated in the APOSTEL 8 trial, therefore, either exposed to atosiban acetate or placebo during pregnancy. We will use four parent-reported questionnaires to assess neurodevelopment, behaviour problems and general health. Data collected in the original trial will be linked to the data obtained in this follow-up study.
Atosiban Acetate Inhibits Prostaglandin F2α-induced Contractions
Oxytocin (OT) is a potent stimulator of myometrial contractions and plays an important role in the initiation of both term and preterm labour. OT stimulates myometrial contractions through multiple signalling pathways. Atosiban Acetate is primarily an arginine vasopressin (AVP) V1a receptor antagonist with lower affinity for the OTR. Its mechanism of action is via dose-dependent inhibition of OT-mediated increase in intracellular calcium levels which involves closing of voltage gated channels to prevent influx of calcium. We aimed to identify the secondary intracellular effects, such as changes in intracellular calcium levels and to assess whether nolasiban and/or atosiban can inhibit PGF2α-induced downstream pro-inflammatory responses in human myometrial smooth muscle cells.[3]
We have shown that atosiban acetate and nolasiban reduced not only OT-induced myometrial contractions in a dose-dependent manner but also PGF2α-induced myometrial contractions in an ex vivo myometrial contractility model. The two OTR-As have comparable potency in suppressing OT- and PG-augmented contractions. Atosiban Acetate is a well-established OTR-A which is approved in Europe. Although the efficacy of atosiban in clinical trials is disputed, it has been repeatedly shown to successfully inhibit OT-induced myometrial contractions in vitro by inhibiting inositol triphosphate (IP3) production with decrease in intracellular calcium mobilisation in the myometrial cell as we have shown in this study. Nolasiban is a non-peptide OTR-A, which has a higher selectivity for OTR than V1a receptors when compared to atosiban acetate. In conclusion, both OTR-As, atosiban and nolasiban, suppress OT- and PGF2α-induced calcium response to dose-dependently inhibit pregnant myometrial contractions.
References
[1]Zhu H, Gu W, Wang B, Hu R. The effectiveness and safety of continuous and repeated treatment of atosiban in twin pregnancy with threatened preterm labor: A propensity score-matched study. PLoS One. 2025 Jul 9;20(7):e0328008. doi: 10.1371/journal.pone.0328008. PMID: 40632745; PMCID: PMC12240366.
[2]van der Windt L, Klumper J, van Limburg Stirum EVJ, van 't Hooft J, van Wely M, van Wassenaer-Leemhuis AG, Pajkrt E, Oudijk MA; APOSTEL 8 Study Group. Atosiban versus placebo in the treatment of threatened preterm birth between 30 and 34 weeks gestation: study protocol of the 4-year APOSTEL 8 follow-up. BMJ Open. 2024 Jul 18;14(7):e083600. doi: 10.1136/bmjopen-2023-083600. PMID: 39025819; PMCID: PMC11261668.
[3]Kim SH, Riaposova L, Ahmed H, Pohl O, Chollet A, Gotteland JP, Hanyaloglu A, Bennett PR, Terzidou V. Oxytocin Receptor Antagonists, Atosiban and Nolasiban, Inhibit Prostaglandin F2α-induced Contractions and Inflammatory Responses in Human Myometrium. Sci Rep. 2019 Apr 8;9(1):5792. doi: 10.1038/s41598-019-42181-2. PMID: 30962532; PMCID: PMC6453954.
You may like
Lastest Price from Atosiban Acetate manufacturers

US $1000.00-1000.00/g2025-09-19
- CAS:
- 914453-95-5
- Min. Order:
- 1g
- Purity:
- >=99% (HPLC), USP43
- Supply Ability:
- 10000000g

US $0.00/g2025-01-13
- CAS:
- 914453-95-5
- Min. Order:
- 1g
- Purity:
- More Than 99%
- Supply Ability:
- 50kg/Month