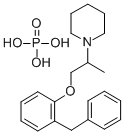
Benproperine Phosphate as an Antitussive Drug
Benproperine phosphate is an orally active, potent actin-related protein 2/3 complex subunit 2 (ARPC2) inhibitor. It attenuates the actin polymerization rate of action polymerization nucleation by impairing Arp2/3 function. Benproperine phosphate has the potential for a cough suppressant and suppresses cancer cell migration and tumor metastasis.

Repurposing antitussive benproperine phosphate against pancreatic cancer
Pancreatic cancer (PC) is one of the most intractable tumors with low survival rate and high mortality. Currently, surgical resection is still generally recognized as the most effective clinical treatment of PC, which causes great suffering to patients regretfully, especially the elderly. Drug repurposing has recently drawn growing attention in the cancer management with favorable therapeutic effects. Several classes of antifungal and antiparasitic agents have shown great potential to be repurposed for anticancer application. Benproperine phosphate (BPP), which is widely used as a nonproductive cough suppressant, has been reported to exhibit anticancer activities in several cancer cells. It has been reported that BPP inhibits cancer metastasis through regulation of actin‐related protein 2/3 complex subunit 2 (ARPC2)‐mediated pathway or inhibition of angiogenesis. In addition, Benproperine phosphate also shows suppressive effects on PC cells both in vitro and in vivo. However, the underlying mechanisms remain poorly understood. In this study, we demonstrate the anticancer effect of BPP for PC treatment both in vitro and in vivo. Interestingly, we find that BPP induces autophagy initiation and autophagosome formation via regulating AMPK/mTOR pathway. Meanwhile, BPP inhibits autophagosome–lysosome fusion by downregulating Ras‐related protein Rab‐11A (RAB11A) level leading to excessive accumulation of autophagosomes and lethal autophagy arrest. Our study reveals a novel anticancer mechanism for Benproperine phosphate and demonstrates the therapeutic potential of BPP for the treatment of PC.[1]
Recently, BPP has been reported to exhibit anticancer effects in a variety of tumor models. In this study, we demonstrated that Benproperine phosphate significantly suppressed cell proliferation in PC both in vitro and in vivo. We also showed that BPP markedly induced autophagy initiation via regulating AMPK/mTOR pathway and subsequently somehow increased the protein levels of Atg5 and Beclin 1 (maybe through enhancing their transcriptional levels or inhibiting their degradation), contributing to autophagy initiation. Combinatorial treatment of autophagic inhibitor 3‐MA or knockdown of autophagy‐related protein by siRNA decreased BPP‐mediated growth inhibition. We further revealed that Benproperine phosphate perturbed the fusion of autophagosomes with lysosomes by reducing the expression of RAB11A, thus resulting in an excessive accumulation of autophagosomes and cell death. Our study implies BPP as a potential candidate for PC treatment. In this study, we demonstrate that BPP inhibits the growth of PC cells both in vitro and in vivo. BPP induces autophagy initiation by the regulation of AMPK/mTOR pathways. Interestingly, BPP disturbs the fusion of autophagosomes with lysosomes by downregulating the expression of RAB11A, thus leading to excessive accumulation of autophagosomes and cell death. Our study suggests Benproperine phosphate as a potential repurposed nononcology drug for PC treatment.
Benproperine phosphate enhances pancreatic Cancer chemotherapy
Pancreatic cancer is one of the most lethal malignancies, with a five-year survival rate of less than 10 %. Currently, chemotherapy remains one of the most effective treatment approaches for pancreatic cancer. In recent years, drug repurposing has emerged as a promising strategy to accelerate the development of anticancer therapies and address the shortage of effective treatments. Our previous study revealed that benproperine phosphate (BPP), a widely used antitussive agent, exhibited significant anti-pancreatic cancer activity both in vitro and in vivo. BPP induced autophagy initiation by regulating the AMPK/mTOR pathway in pancreatic cancer. Furthermore, Benproperine phosphate inhibited autophagosome-lysosome fusion by downregulating Ras-related protein in brain 11 A (RAB11A), therefore leading to lethal autophagy arrest. In contrast to CQ, which merely blocks autophagosome-lysosome fusion, this dual mechanism of BPP allows to convert Gem-induced protective autophagy into lethal autophagy, markedly improving chemotherapeutic outcomes.[2]
Gemcitabine-based chemotherapy remains a cornerstone in pancreatic cancer treatment, yet its efficacy is hindered by poor bioavailability and adaptive resistance mechanisms, such as autophagy. In this study, we developed a hyaluronic acid (HA) modified zeolitic imidazolate framework-8 (ZIF-8) drug-repurposing nanoplatform (HA/ZIF-8@BPP/Gem) against pancreatic cancer through the co-delivery of the antitussive benproperine phosphate and gemcitabine (Gem). Using cell lines, patient-derived xenograft models, and orthotopic tumor models, we demonstrated that BPP and Gem, rapidly released from the nanoplatform in the acidic tumor microenvironment, exhibited synergistic cytotoxicity without causing significant biochemical abnormalities or organ toxicity. Mechanistically, Benproperine phosphate initiated autophagy but blocked RAB11A-dependent autophagosome-lysosome fusion, thereby converting Gem-induced protective autophagy into a lethal process. In addition, RNA sequencing and flow cytometry analyses revealed that HA/ZIF-8@BPP/Gem stimulated the secretion of immune-related cytokines and activated immune response pathways, particularly T cell-mediated immunity. In conclusion, our study presents a safe and effective strategy of nano-enabled drug repurposing to improve chemotherapy outcomes by inducing amplified autophagy arrest and immune activation in pancreatic cancer.
References
[1]Zhang H, Zhang Z, Huang Y, Qin S, Zhou L, Weng N, Liu J, Yang M, Zhang X, Lu Y, Ma L, Zheng S, Li Q. Repurposing antitussive benproperine phosphate against pancreatic cancer depends on autophagy arrest. Mol Oncol. 2021 Feb;15(2):725-738. doi: 10.1002/1878-0261.12854. Epub 2020 Dec 12. PMID: 33226737; PMCID: PMC7858282.
[2]Zhang Z, Chen Q, Zheng R, Shao Z, Wei X, Gu X, Zang Y, Yang X, Wu J, Dai M, Cao W, Wang Y, Zhang Y, Hu C, Chen C, Wu L, Liang T, Zhang Q. Nano-enabled repurposing of Benproperine phosphate enhances pancreatic Cancer chemotherapy through lethal autophagy arrest and immune activation. J Control Release. 2025 Nov 10;387:114187. doi: 10.1016/j.jconrel.2025.114187. Epub 2025 Sep 1. PMID: 40902670.
You may like
See also
Lastest Price from Benproperine phosphate manufacturers
US $0.00-0.00/kg2025-04-15
- CAS:
- 19428-14-9
- Min. Order:
- 1kg
- Purity:
- 99.0%
- Supply Ability:
- 10tons

US $100.00-1.00/kg2024-03-22
- CAS:
- 19428-14-9
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20 tons