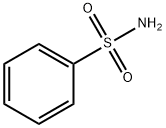
Benzenesulfonamide: Synthetic Applications & Bioactive Derivative Development
Benzenesulfonamide is an inhibitor of human carbonic anhydrase B. Its derivatives are effective in the treatment of proliferative diseases such as cancer. It is used in the synthesis of dyes, photochemicals and disinfectants. It is also used in the synthesis of dyes, photochemistry and disinfectants. In chemical laboratories, it serves as a useful reagent. It participates in numerous chemical reactions, helping to transform one compound into another. It can act as a catalyst or a reactant, enabling chemists to create complex organic molecules. Whether it's for developing new materials or conducting research, benzenesulfonamide plays a key role in facilitating these chemical transformations. It also contributes to creating vibrant and long-lasting colors. It helps improve the colorfastness of dyes, meaning they won't fade easily when exposed to sunlight, water, or washing. This is important for ensuring the quality and appearance of colored products such as textiles, paints, and printing inks.

New benzenesulfonamide derivatives as anticancer and antimicrobial agents
Cancer remains one of the leading causes of death worldwide, thus necessitating the development of novel and effective therapies. Although modern therapeutic agents have been developed over the past 100 years, the successful treatment of cancer appears to be a powerful challenge at the start of the present century. This challenge lies in the difficulty to discover novel selective agents that inhibit the proliferation of tumor cells without being toxic to normal cells. A series of diamide-based benzenesulfonamides were synthesized and evaluated as inhibitors of the metalloenzyme CA IX, compound III achieved the best CA IX/I and IX/II selectivity with selective indexes (SIs) of 985 and 13.8, respectively. SLC-0111 and some of its analogues IVa–d are among isoform-selective sulfonamides that are discovered as CAIs in the last period. From previously mentioned, subsequently, we represent the synthesis, and biological evaluation of a new series of 4-thiazolone-based benzenesulfonamides 4a–j with the “tail approach”. It is based on appending “tails”, with a different chemical nature, to the aromatic or heterocyclic ring owning the zinc binding sulfonamide group, to interact with the middle and the rim parts of the active site cavity. Moreover, bioisosteric replacement was applied by using benzylidene tails incorporating different substituents to ensure appropriate SAR exploration concerning the hydrophobic region of the binding cleft.[1]
The inhibitory potency of all the synthesized thiazol-4-one-benzenesulfonamide derivatives against carbonic anhydrase IX was evaluated. The results represented as IC50 values and are summarized. CA IX activity was carried out to all the synthesized compounds to determine their inhibitory abilities to decrease the reaction of hydrogenocarbonate formation and to further gain insight into structure activity relationships. Investigation of the results of CA IX inhibitory activities among the synthesized compounds revealed that the inhibitory activity ranges from 10 to 338 nM. In particular, substituent with the eugenol aldehyde was well tolerated and the best result was obtained in compound 4e (IC50 = 10.93 nM). In order to go in deep sight to the selectivity of the benzenesulfonamides derivatives, CA II enzyme inhibition was carried out to the most potent derivatives 4e, 4g and 4h. The results revealed that the synthesized compounds didn't inhibit the cytosolic CA II which is a required feature for compounds targeting tumor – associated enzymes. This study was conducted to identify the activity of the newly synthesized benzenesulfonamide derivatives and to understand some structural observations for their binding modes and their binding interactions with CA IX enzyme. The 3D crystal structure of CA IX (PDB ID: 5FL6) in complex with 5-(1-(4-methylphenyl)-1H-1,2,3-triazol-4-yl)thiophene-2-sulfonamide was used for this docking study.
Identification of Novel and Potent Indole-Based Benzenesulfonamides
Carbonic anhydrases (CAs) are metalloenzymes found in all living things that are required for the conversion of CO2 to bicarbonate and protons. CAs observed in humans belong to the α-class (α-CA), and are classified into sixteen isoforms that vary in molecular characteristics, oligomeric structure, cellular localization, distribution in organs and tissues, expression levels, and kinetic properties. The distribution pattern of the CA isoforms can be described as, cytosolic (I, II, III, VII, and XIII), membrane (IV, IX, XII, and XIV), secretory (VI), and mitochondrial (VA and VB) forms. Inspired by all these efforts related to the development of indole-based hCA inhibitors, we designed a novel series of indole-based benzenesulfonamides through a hybridization process of these two scaffolds. Thereafter, a regioisomerism of the ZBG moiety was carried out to afford fifteen indole-based benzenesulfonamides linked together via an amide linker, giving the synthesized compounds flexibility as well as the ability to form hydrogen bond interactions with the target protein. To the best of our knowledge, this hybridization approach of the indole moiety with the most effective ZBG moiety (the benzenesulfonamide group) via a flexible amide linker on different positions on the indole core to develop novel small molecules with a promising potential to inhibit carbonic anhydrase isoforms has not been reported so far.[2]
In recent decades, human carbonic anhydrase inhibitors (hCAIs) have emerged as an important therapeutic class with various applications including antiglaucoma, anticonvulsants, and anticancer agents. Herein, a novel series of indole-based benzenesulfonamides were designed, synthesized, and biologically evaluated as potential hCAIs. A regioisomerism of the sulfonamide moiety was carried out to afford a total of fifteen indole-based benzenesulfonamides possessing different amide linkers that enable the ligands to be flexible and develop potential H-bond interaction(s) with the target protein. The activity of the synthesized compounds was evaluated against four hCA isoforms (I, II, IX and, XII). Among all, compound 2a demonstrated the most potent inhibition against the hCA II isoform with an inhibitory constant (Ki) of 5.9 nM, with 13-, 34-, and 9-fold selectivity for hCA II over I, IX and XII isoforms, respectively. Structure–activity relationship data attained for various substitutions were rationalized. Furthermore, a molecular docking study gave insights into both inhibitory activity and selectivity of the target compounds. Accordingly, this report presents a successful scaffold hoping approach that reveals compound 2a as a highly potent and selective indole-based hCA II inhibitor worthy of further investigation.
Synthesis and Antidiabetic Evaluation of Benzenesulfonamide Derivatives
In continuation of our research program to develop small molecules as biologically active compounds, in this paper we report the synthesis and structural characterization of several benzenesulfonamides derivatives. These compounds were evaluated for their hypoglycemic activity after administration at dose of 100 mg/Kg in Alloxan-STZ induced diabetic rat. Blood glucose level were measured and compared with control drug, Glibenclamide (5 mg/Kg) as a standard. A series of N-(4-phenylthiazol-2-yl) benzenesulfonamides derivatives were synthesized by reacting of equimolar quantities of 2-amino thiazol (compound 3 and 4) with appropriate aromatic sulfonyl chloride. The structures of these compounds were established by means of IR, 1H NMR, and elemental analysis. All the compounds were screened in-vivo for their oral hypoglycemic activity by streptozotocin-induced diabetic model in rat. Four of the compounds demonstrated remarkable hypoglycemic property, however with a degree of variation. The results of changes in blood glucose in diabetic rats treated with 100 mg/Kg p.o. of the synthesized benzenesulfonamides derivatives are presented.[3]
This study reports the synthesis and antidiabetic activity of novel N-(4-phenylthiazol-2-yl) benzenesulfonamides derivatives. Some of the synthesized compounds showed hypoglycemic activity. These results indicated that benzenesulfonamide could be served as potential antidiabetic agents in the same manner as sulfonylurea derivatives. It seems that structural modification of this scaffold will resulted in more potent oral antidibetic derivatives as it will be followed in our future projects.
References
[1]Nemr MTM, AboulMagd AM, Hassan HM, Hamed AA, Hamed MIA, Elsaadi MT. Design, synthesis and mechanistic study of new benzenesulfonamide derivatives as anticancer and antimicrobial agents via carbonic anhydrase IX inhibition. RSC Adv. 2021 Aug 1;11(42):26241-26257. doi: 10.1039/d1ra05277b. PMID: 35479426; PMCID: PMC9037358.
[2]Elkamhawy A, Woo J, Nada H, Angeli A, Bedair TM, Supuran CT, Lee K. Identification of Novel and Potent Indole-Based Benzenesulfonamides as Selective Human Carbonic Anhydrase II Inhibitors: Design, Synthesis, In Vitro, and In Silico Studies. Int J Mol Sci. 2022 Feb 25;23(5):2540. doi: 10.3390/ijms23052540. PMID: 35269684; PMCID: PMC8910009.
[3]Hosseinzadeh N, Seraj S, Bakhshi-Dezffoli ME, Hasani M, Khoshneviszadeh M, Fallah-Bonekohal S, Abdollahi M, Foroumadi A, Shafiee A. Synthesis and antidiabetic evaluation of benzenesulfonamide derivatives. Iran J Pharm Res. 2013 Spring;12(2):325-30. PMID: 24250607; PMCID: PMC3813231.
You may like
See also
Lastest Price from Benzenesulfonamide manufacturers

US $2.00-6.00/kg2025-07-29
- CAS:
- 98-10-2
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 100kg

US $1.00/kg2025-04-21
- CAS:
- 98-10-2
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 10 mt