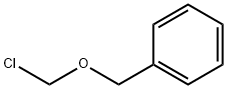
Benzyl Chloromethyl Ether: Synthesis & Alkylation Applications
Benzyl chloromethyl ether is useful for introduction of a potential hydroxymethyl group in alkylation reactions. Hill and Keach first used this method and found it convenient in barbiturate syntheses. Graham and McQuillin, and Graham, McQuillin, and Simpson12 have extended the scope of the alkylation reaction to various ketone derivatives. They also have investigated the conditions for obtaining maximum C-alkylation and the stereochemistry of alkylation in various octalone systems. Alkylation of ketones followed by sodium borohydride reduction and catalytic hydrogenolysis represents a convenient method for obtaining 1,3-diols. Similarly, Wolff-Kishner reduction and catalytic hydrogenolysis give primary alcohols. A procedure of this type has been used for obtaining bridgehead methanol derivatives of bicyclic compounds. Alkylation of ester enolates, generated by lithium diisopropylamide, has been reported. Benzyl chloromethyl ether has been prepared from benzyl alcohol, aqueous formaldehyde solution, and hydrogen chloride. Gaseous formaldehyde and 1,3,5-trioxane have also been used. This chloromethyl ether has also been prepared by the chlorination of benzyl methyl ether. The present procedure is based on the first method, but avoids the use of a large excess of formaldehyde and provides a considerably simplified isolation procedure.

In Situ Synthesis of Benzyl Chloromethyl Ether
In recent years the synthesis and biological screening of -peptides have been widely applied. The methodology here described is developed for the synthesis of hexapeptides analog of neurotensin NT Arg-Arg-Pro Tyr-Ileu-Leu-OH. We wish to report herein the in-situ synthesis of benzyl chloromethyl ether and its use to protect the hydroxyl functionality in various bifunctional organic com OH 1 (CH2O)n H2SO4 Azeotroic distillation Benzene chloride in catalytic amount of dry benzyl alcohol at room temperature under static pressure of nitrogen for 36 h to afford benzyl chloromethyl ether, which was ready to use without any work up. The general procedure to protect the hydroxyl functional ity of organic molecule is as follow. Hydroxyl compound (1 mmol) was dissolved in refluxing dried chloroform under static pressure of nitrogen. To this solution benzyl chloro methyl ether (3 mmol) and diisopropylethyl amine (4 mmol) were added subsequently, refluxing for 8 h. The reaction mixture was cooled to room temperature, diluted with pounds as well as its deprotection in the presence of other acid sensitive protecting groups. Formaldehyde dibenzylacetal was synthesized by azeotropic distillation of the mixture of benzyl alcohol, paraformaldehyde, and catalytic amount of concentrated sulfuric acid in dry benzene, however, use of toluene as a solvent does not affect the yield of product.[1]
Benzyloxymethyl group is an attractive protecting group in the sense that it can easily be re moved in mild acidic as well as in neutral conditions through catalytic hydrogenation, while benzyl chloromethyl ether is highly toxic. Though this method seems to be attrac- tive to synthesize benzyloxymethyl ester of N-protected L- amino acids because they can be deprotected under mild acidic as well as in neutral conditions using catalytic hydro- genation, however, we still have some reservations about this method. HMPA is highly carcinogenic while it is hard to get sodium salts of N-protected L-amino acids. Therefore we applied the conditions as previously described to the esterifi- cation of N-protected L-amino acids. Benzyloxymethyl es- ters of various N-protected L-amino acids (18) were obtained in moderate yields. In this way we could avoid the use of carcinogenic reagents HMPA and benzyl chloromethyl ether without using sodium salts of N-protected L-amino acids. In conclusion in situ synthesis of benzyl chloromethyl ethers provides an easy excess to BOM protection of hy droxyl functionality of alkyl and aryl alcohols with the extra advantage of orthogonality with t-butyldimethylsilyl group. In addition by using this method secondary alcohol can be deprotected in the presence of primary one.
References
[1]Letters in Organic Chemistry. 2008;5(2):125-127. doi:10.2174/157017808783743894
You may like
See also
Lastest Price from Benzyl chloromethyl ether manufacturers

US $0.00/KG2025-04-15
- CAS:
- 3587-60-8
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 500000kg

US $100.00-32.00/kg2025-03-07
- CAS:
- 3587-60-8
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 5 tons per month