Besifloxacin hydrochloride:Crystal structure and Determination methods of enantiomeric impurity
Introduction
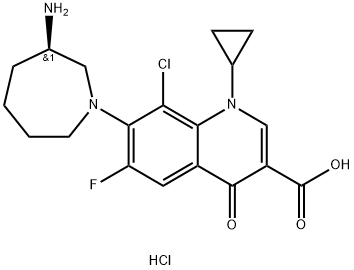
Besifloxacin hydrochloride (Figure.1) is a fourth-generation fluoroquinolone-type ophthalmic antibiotic for the treatment of bacterial conjunctivitis.It was approved by the FDA on May 28, 2009. The systematic name (CAS Registry Number 405165-61-9) is 7-[(3R)-3-aminoazepan-1-yl]-8-chloro-1-cyclopropyl-6-fluoro-4-oxoquinoline-3-carboxylic acid hydrochloride. Besifloxacin free base is claimed as a treatment for ophthalmic infections in US Patent 8,415,342, but no X-ray powder diffraction data are provided. US Patent 8,481,526 claims a molecular crystal form of (R)-(+)-besifloxacin with X-ray powder diffraction peaks at 10.6, 15, 19.7, 21.1, and 22±0.2°2θ(Cu radiation), and powder patterns for both the free base and hydrochloride salt are provided. Similar diffraction patterns are also contained in US Patent 8,604,020. An Australian Department of Health report (2013) notes that besifloxacin hydrochloride may exist as a racemic mixture of the R-(+) and S-(−) enantiomers; although both enantiomers exhibit antibacterial activity,the active limit for a proposed ophthalmic product is the R-enantiomer with a limit on the S-enantiomer of 0.5 wt%.[1]

Crystal structure of besifloxacin hydrochloride
The crystal structure of besifloxacin hydrochloride has been solved and refined using synchrotron X-ray powder diffraction data, and optimized using density functional theory techniques. Besifloxacin hydrochloride crystallizes in space group P1 (#1) with a = 5.36596(8), b=10.3234(4), c=17.9673(14) Å, α=98.122(5), β=92.9395(9), γ=96.1135(3)°, V=977.483(13) Å3, and Z=2. The crystal structure is approximately centrosymmetric. Strong N–H⋯Cl hydrogen bonds form a corrugated ladder-like chain along the a-axis. The carboxylic acid group in each independent cation acts as the donor in a strong intramolecular O–H⋯O hydrogen bond to an adjacent carbonyl group. The powder pattern has been submitted to ICDD for inclusion in the Powder Diffraction File™ (PDF®).[1]
Analysis of Organic Impurities of Besifloxacin Hydrochloride by HPLC
Impurity analysis is an important step in the quality control of pharmaceutical ingredients and the final product. Impurities can arise from drug synthesis or excipients and even at small concentrations may affect product efficacy and safety. In this work, two methods using highperformance liquid chromatography (HPLC) were developed and validated for the evaluation of besifloxacin hydrochloride and its impurity synthesis, with isocratic elution and another with gradient elution.;Method: The analysis by HPLC in isocratic elution mode was performed using a cyano column maintained at 25 °C. The mobile phase was composed of 0.5% triethylamine (pH 3.0): acetonitrile (88:12 v/v) eluted at a flow rate of 1.0 mL/min with detection at 330 nm. The gradient elution method was carried out with the same column and mobile phase components only modifying the rate between organic and aqueous phase during analysis. The procedures have been validated according to internationally accepted guidelines, observing results within acceptable limits. The methods presented were found to be linear in the 140 to 260 μg/ mL range for besifloxacin hydrochloride and 0.3 to 2.3 μg/ mL for an impurity named A. The limits of detection and quantification were, respectively, 0.07 and 0.3 μg/ mL for impurity A, with a 20 μL injection volume. The precision achieved for all analyses performed provided RSD inter-day equal to 6.47 and 6.36% for impurity A with isocratic elution and gradient, respectively. The accuracy was higher than 99% and robustness exhibited satisfactory results. In the isocratic method, an analysis time of 25 min and 15 min was obtained for the gradient. For impurity A, the number of theoretical plates in the isocratic mode was about 5000 while in the gradient mode it was about 45000, hence, it made the column more efficient by changing the mobile phase composition during elution. In besifloxacin hydrochloride raw material and in the pharmaceutical product used in this study, other related impurities were present but impurity A was searched for and not detected. The proposed methods can be applied for the quantitative determination of impurities in the analysis of the besifloxacin hydrochloride raw material, as well as in ophthalmic suspension of the drug, considering the quantitation limit.[2]
Determination of enantiomeric impurity in besifloxacin hydrochloride by chiral HPLC
Besifloxacin hydrochloride is a novel chiral broad-spectrum fluoroquinolone developed for the treatment of bacterial conjunctivitis. R-besifloxacin hydrochloride is used in clinics as a consequence of its higher antibacterial activity. To establish an enantiomeric impurity determination method, some chiral stationary phases (CSPs) were screened. Besifloxacin enantiomers can be separated to a certain extent on Chiral CD-Ph (Shiseido Co., Ltd., Japan), Chiral AGP, and Crownpak CR (+) (Daicel Chemical IND., Ltd., Japan). However, the selectivity and sensitivity were both unsatisfactory on these three CSPs. Therefore, Chiral AGP, Chiral CD-Ph, and Crownpak CR (+) were not used in the enantiomeric impurity determination of besifloxacin hydrochloride. The separation of enantiomers of besifloxacin was further performed using a precolumn derivatization chiral high-performance liquid chromatography method. 2,3,4,6-Tetra-O-acetyl-beta-D-glucopyranosyl isothiocyanate was used as the derivatization reagent. Besifloxacin enantiomer derivates were well separated on a C(18) column (250 × 4.6 mm, 5 µm) with a mobile phase that consisted of methanol-KH(2)PO(4) buffer solution (20 mM; pH 3.0) (50:50, v/v). Selectivity, sensitivity, linearity, accuracy, precision, stability, and robustness of this method were all satisfied with the method validation requirement. The method was suitable for the quality control of enantiomeric impurity in besifloxacin hydrochloride.[3]
Determination of Enantiomeric Impurity in Besifloxacin Hydrochloride
Besifloxacin is a unique chiral broad-spectrum flouroquinolone used in the treatment of bacterial conjunctivitis. R-form of besifloxacin hydrochloride shows higher antibacterial activity as compared to the S-isomer. Therefore, it is necessary to establish chiral purity. To establish chiral purity a high-performance liquid chromatography (HPLC) method for determination of R-besifloxacin and S-besifloxacin (BES impurity A) was developed and validated for in-process quality control and stability studies. The analytical performance parameters such as linearity, precision, accuracy, specificity, limit of detection (LOD), and lower limit of quantification (LOQ) were determined according to International Council for Harmonization ICH Q2(R1) guidelines. HPLC separation was achieved on Chiralpak AD-H (250 x 4.6 mm, 5 μm) column using n-heptane: ethanol: ethylenediamine: acetic acid (800:200:0.5:0.5) (v/v/v/v) as the mobile phase in an isocratic elution. The eluents were monitored by UV/Visible detector at 290 nm. The resolution between S-isomer and besifloxacin hydrochloride was more than 2.0. Based on a signal-to-noise ratio of 3 and 10 the LOD of besifloxacin was 0.30 μg/mL, while the LOQ was 0.90 μg/mL. The calibration curves were linear in the range of 0.9-7.5 μg/mL. Precision of the method was established within the acceptable range. The method was suitable for the quality control enantiomeric impurity in besifloxacin hydrochloride. [4]
References
[1] A. J K ,Stacy G ,N. T B .Crystal structure of besifloxacin hydrochloride, C19H22ClFN3O3Cl[J].Powder Diffraction,2023,38(1):43-52.DOI:10.1017/S0885715622000586.
[2] [1]AlvesGiordani FC ,Garcia VC ,ShermanSchapoval EE , et al.Analysis of Organic Impurities of Besifloxacin Hydrochloride by High-Performance Liquid Chromatography with Isocratic and Gradient Elution[J].Current Pharmaceutical Analysis,2021,17(4):564-572.
[3] Wang Z, Wang S, Zhu F, Chen Z, Yu L, Zeng S. Determination of enantiomeric impurity in besifloxacin hydrochloride by chiral high-performance liquid chromatography with precolumn derivatization. Chirality. 2012;24(7):526-531. doi:10.1002/chir.22042
[4] Kumar GP, Srivastava V, Khandelwal K, et al. Simple Isocratic HPLC Method for Determination of Enantiomeric Impurity in Besifloxacin Hydrochloride. Chirality. 2016;28(9):628-632. doi:10.1002/chir.22626
You may like
Related articles And Qustion
See also
Lastest Price from Besifloxacin hydrochloride manufacturers
US $0.00/kg2025-11-21
- CAS:
- 405165-61-9
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- Customise

US $0.00/Kg/Drum2025-04-21
- CAS:
- 405165-61-9
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 500kg