Bioactivity and Animal experiments of Retinal
Retinal, also known as vitamin A aldehyde, is an oxidation derivative of retinol. Under normal conditions, it appears as an orange crystalline solid, exhibits notable sensitivity to oxidants, and is prone to oxidative degradation upon exposure to strong oxidizing agents. Retinal is insoluble in water but soluble in ethanol, chloroform, cyclohexane, petroleum ether, and oils. It was first isolated from the retina and can be generated from the oxidative cleavage of β-carotene. Its physiological effects are related to vision; it is generally recognized to undergo rapid cis-trans isomerization upon light exposure.

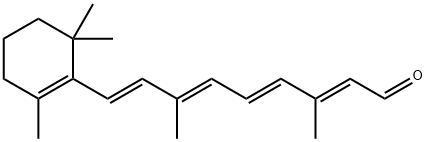
Figure1: Picture of Retinal
Introduction
Within visual cells, 11-cis-retinal combines with opsin to form visual pigments. Upon absorbing light, 11-cis-retinal isomerizes into Retinal, inducing conformational changes in rhodopsin that initiate neural impulses to the brain, thereby generating vision. During the breakdown and resynthesis of rhodopsin, a portion of retinal is consumed, primarily replenished by vitamin A from the bloodstream. Retinal is one of the major vitamin A metabolites in the retina, produced after photoisomerization of the visual chromophore 11-cis-retinal. All-Retinal is cleared from photoreceptors by ATP-binding cassette transporters (ABCA4) and all-trans-retinol dehydrogenase (RDH). Additionally, all-Retinal can mediate retinal cell apoptosis by activating Bax via DNA damage. Within the rod cells of the retina, 11-cis-retinal is present and, upon exposure to light, is converted into all-Retinal. This transformation triggers the breakdown of rhodopsin, thereby generating visual perception. Thus, it is evident that the all-Retinal molecule is closely intertwined with our daily lives. [1]
Bioactivity
Retinal is an vitamin A metabolite in the retina, and is produced following photo-isomerization of the visual chromophore 11-cis-Retinal. All-Retinal is cleared from photoreceptors by ATP-binding cassette transporter (ABCA4) and all-trans-retinol dehydrogenase (RDH). All-Retinal induces Baxactivation via DNA damage to mediate retinal cell apoptosis. Retinal is an important signaling molecule in eye development, playing multiple critical roles in the ocular development of vertebrates. Myopia is a developmental disorder in which active scleral expansion serves as a key mechanism for axial elongation. Retinaldehyde (retinal) may function as a messenger molecule regulating experimental myopia progression. Research on the relationship between trans‑Retinal and the onset and progression of experimental myopia has achieved notable progress. Studies have reported on trans‑Retinal and its nuclear receptors, alterations in retinaldehyde levels in the retina, choroid, and sclera of experimental myopic eyes, as well as advances in understanding retinaldehyde as a messenger transmitting retinal signals to the sclera to mediate ocular elongation. Retinoic acid is widely used in dermatology, but its clinical application is partly limited due to local irritation. trans‑Retinal, as a natural intermediate metabolite of retinoic acid, exhibits biological activities similar to retinoic acid while demonstrating significantly better skin tolerability.
Animal experiments
The visual (retinoid) cycle is a fundamental metabolic process in the vertebrate retina responsible for producing 11‑cis‑retinal, the chromophore of rhodopsin and cone pigments. 11‑cis‑Retinal binds to opsins, forming visual pigments. When the resulting visual chromophore, 11‑cis‑retinylidene, is photoisomerized to trans‑retinylidene, trans‑retinal is released from these receptors. Toxic byproducts derived from **trans**‑retinal during the visual cycle are frequently associated with lipofuscin deposits in the retinal pigment epithelium (RPE). However, it remains unclear whether aberrant reactions of the visual cycle contribute to RPE atrophy, which may trigger rapid-onset retinopathy. It has been reported that mice lacking both ATP‑binding cassette transporter 4 (Abca4) and retinol dehydrogenase 8 (Rdh8)—proteins essential for clearing trans‑retinal from photoreceptors—develop severe RPE/photoreceptor dystrophy at an early age. This phenotype includes lipofuscin accumulation, drusen, basal laminar deposits, Bruch’s membrane thickening, and choroidal neovascularization. Importantly, the severity of visual dysfunction and retinopathy was exacerbated by light exposure but attenuated by treatment with retinylamine, a visual-cycle inhibitor that slows the flux of trans‑retinal through the cycle. These findings provide direct evidence that aberrant production of toxic condensation byproducts from the visual cycle in mice can lead to rapid, progressive retinal degeneration. [2]
Reference
[1] Balke D E, Becker R S. Spectroscopy and photochemistry of all-trans-retinal and 11-cis-retinal[J]. Journal of the American Chemical Society, 1967, 89(19): 5061-5062.
[2] Maeda A, Maeda T, Golczak M, et al. Retinopathy in mice induced by disrupted all-trans-retinal clearance[J]. Journal of Biological Chemistry, 2008, 283: 26684-26693.
You may like
See also
Lastest Price from Retinal manufacturers

US $0.00-0.00/kg2026-02-10
- CAS:
- 116-31-4
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 10 tons

US $0.00-0.00/kg2026-02-10
- CAS:
- 116-31-4
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- Ex:20 tons