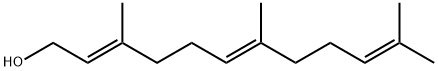
Biological Activity and Application of (E,E)-Farnesol
(E,E)-Farnesol is a naturally occurring molecule and serves as one of the key bioactive constituents in traditional Chinese medicinal plants. Typically appearing as a colorless, transparent liquid, (E,E)-farnesol possesses a mild lily-of-the-valley floral scent and is miscible with water as well as most organic solvents. This compound is found in the essential oils extracted from flowers, leaves, and seeds of certain plants and finds applications in perfumery, antibacterial and insecticidal formulations, and pharmaceutical development.

Figure1: Picture of (E,E)-farnesol
Structural properties
(E,E)-farnesol, as a colorless oily liquid, possesses a pleasant, mild, and delicate floral aroma with characteristics of lily of the valley. Due to its excellent fixative properties, it is commonly used as a base note and serves as an important fragrance ingredient in high-end floral perfumes. The physical constants of (E,E)-farnesol may vary depending on its source and isomers. Since the farnesol molecule contains three trans double bonds, it is recommended to store it at low temperatures with proper sealing to effectively minimize oxidation.
Synthesis
A synthetic route for (E,E)-farnesol (trans,trans‑farnesol, also known as (2E,6E)‑farnesol) has been disclosed in the public literature. The described four‑step procedure starts from geranyl acetate and proceeds sequentially through selenium dioxide/tert‑butyl hydroperoxide oxidation, acid‑catalyzed acetal formation with dihydropyran, strong‑base saponification, and coupling with isopentenyl Grignard reagent in the presence of a copper salt. This reported method for preparing (E,E)-farnesol employs readily available starting materials, features simple operations and mild reaction conditions, and shows promise for industrial‑scale production. [1]
Biological Activity
(E,E)-farnesol is a sesquiterpene acyclic alcohol synthesized by bacteria, protozoa, fungi, plants, and animals. Previous literature has documented its applications in the food, pharmaceutical, and cosmetic industries, as well as its pharmacological role as a vasorelaxant. Nevertheless, its effects on human umbilical vessels remain insufficiently studied. Researchers therefore sought to investigate the vasorelaxant effect of (E,E)-farnesol in human umbilical veins (HUV) from healthy donors using a novel methodological approach. Isolated HUV rings were suspended in an organ bath to record isometric tension across different experimental conditions. (E,E)-Farnesol, at concentrations ranging from 1 μmol/L to 1 mmol/L, induced vasorelaxation in venous preparations pre-contracted either by depolarization (KCl 60 mmol/L) or pharmacological agonism (5-HT 10 μmol/L), with respective EC₅₀ values of 239.9 μmol/L and 424 μmol/L. This relaxant effect was also observed in calcium-free medium. Furthermore, (E,E)-farnesol suppressed contractions evoked by CaCl₂ and BaCl₂, suggesting an inhibitory action on voltage-dependent—particularly L-type—calcium channels. The vasorelaxant efficacy and potency of (E,E)-farnesol were modulated in the presence of tetraethylammonium (1 and 10 mmol/L), glibenclamide (10 μmol/L), and BaCl₂ (1 mmol/L), indicating the potential involvement of potassium channels—such as BKCa, KATP, and KIR—in the observed response. These findings imply that (E,E)-farnesol holds promise as a potential vasodilator for managing hypertensive conditions during pregnancy that are associated with altered HUV reactivity. (E,E)-farnesol is a constituent of essential oils known for their vasodilator properties.
Vasorelaxant effects
Previous studies have demonstrated that (E,E)-farnesol possesses vasorelaxant effects, including in human resistance arteries, as well as hypotensive potential. Collectively, this evidence suggests that (E,E)-farnesol may play a regulatory role within the cardiovascular system. Despite scientific advancements, the direct influence of clinically used synthetic drugs for gestational hypertensive syndromes on the human umbilical venous circulation remains relatively under-reported in in vitro and ex vivo studies. Furthermore, research on the vasoactive effects of natural products, despite their promising pharmacological potential, is even more limited. The human umbilical vein (HUV) is a unique and vital blood vessel for fetal life, responsible for oxygenation and nutrition. Lacking innervation, its vascular tone is regulated by mechanical factors, the local environment, and vasoactive substances from the systemic circulation. Under pathological hypertensive and metabolic conditions during pregnancy, the HUV exhibits heightened responsiveness to abnormal microenvironmental stimuli and undergoes morphological and ultrastructural alterations, phenomena which can compromise intrauterine fetal viability. Current knowledge regarding the effects of (E,E)-farnesol on human umbilical vessel reactivity remains confined to studies on umbilical arteries. Therefore, considering the potential obstetric therapeutic implications of (E,E)-farnesol on umbilical circulation, this study aimed to investigate the vasorelaxant effect of (E,E)-farnesol on umbilical veins obtained from healthy donors.[2]
Effect in human umbilical arteries
(E,E)-Farnesol, a sesquiterpene alcohol derived from plants and animals, exhibits cardiovascular pharmacological properties. Nevertheless, its effects on human umbilical vessels have remained unexplored. To address this gap, researchers aimed to characterize the vasodilatory effect of (E,E)-farnesol on human umbilical arteries (HUA). Umbilical artery tissues were obtained from normotensive pregnant women over 18 years of age with no prepartum complications. After collection, the tissues were segmented, dissected to remove Wharton’s jelly, and prepared as isolated arterial rings for isometric tension recording in an organ bath system. The results demonstrated that (E,E)-farnesol (1 μmol/L to 1 mmol/L) elicited vasodilation in HUA preparations, reducing basal tone and inhibiting both electromechanical coupling induced by KCl (60 mmol/L) and pharmacomechanical coupling induced by 5‑HT (10 μmol/L), with greater potency against KCl‑induced contraction (EC₅₀ 225.3 μmol/L) compared to 5‑HT‑induced contraction (EC₅₀ 363.5 μmol/L). In calcium‑free medium, pharmacomechanical coupling was abolished, and contractions triggered by CaCl₂ or BaCl₂ were attenuated by (E,E)-farnesol, suggesting a direct inhibitory effect on L‑type voltage‑operated calcium channels as a key mechanism underlying its vasodilatory action. Further analysis indicated that the vasodilator efficacy of (E,E)-farnesol was modulated in the presence of tetraethylammonium (1 or 10 mmol/L), 4‑aminopyridine (1 mmol/L), and glibenclamide (10 μmol/L), implying potential involvement of potassium channels—including BKCa, Kᵥ, and KATP—in its vascular effect. [3]
Application
(E,E)-Farnesol is widely distributed in various plant parts such as flowers, leaves, and stems, with notably high concentrations found in certain medicinal herbs and aromatic plants. Plants like the farnesol‑rich variety of Cinnamomum tenuipilum, Magnolia flower buds, and loquat leaves represent promising resources for further development and utilization. As one of the key bioactive constituents in traditional medicinal herbs and a major aromatic component in the essential oils of several important fragrant plants, (E,E)-farnesol has already found extensive applications—and shows continued potential—in fields that leverage its biological activities, including pharmaceuticals, agrochemicals, cosmetics, and daily‑use chemical products.
Preparation of insecticidal composition
Researchers have reported that (E,E)-Farnesol can be used to prepare insecticidal compositions containing this compound. A farnesol-containing insecticidal composition is characterized in that its active ingredients consist of a binary combination of farnesol with one of the following: abamectin, butene-fipronil, cycloxaprid, or pymetrozine. Due to the distinct insecticidal mechanisms of (E,E)-Farnesol and the other active components, their combined application can effectively delay the development of pest resistance and extend the operational lifespan of the insecticidal formulation. Furthermore, in the synthesized insecticidal compositions based on (E,E)-Farnesol, the combination of active ingredients exhibits a synergistic effect against aphids, leading to improved control efficacy, reduced dosage of active compounds, and lower pest management costs. This approach also provides support for the integrated management of aphids and the development of novel chemical agents for aphid control. [4]
Synergistic aphicidal effect
Researchers used Aphis citricola as the test insect, with indoor toxicity assays conducted via the micro-droplet method and field efficacy trials performed according to national standards, to evaluate the combined aphicidal activity of (E,E)-farnesol and nicotine, aiming to provide a foundation for developing novel botanical aphicides. The results showed that (E,E)-farnesol exhibited significant synergistic aphicidal effects when mixed with nicotine, achieving a co-toxicity coefficient (CTC) of 151.63 at the optimal mass ratio (farnesol : nicotine = 4.82 : 1). Through screening of solvents, surfactants, and other adjuvants, a 6% nicotine · farnesol soluble concentrate was successfully developed. [5]
References
[1] Zeng, Q.; Ma, C.; Zhou, L. A Synthesis Method of trans,trans-Farnesol: CN201610233773.3 [P].
[2] Batista, P. R.; de Alencar Silva, A.; de Sena Bastos, C. M.; et al. Vasorelaxant Effect of (E,E)-Farnesol in Human Umbilical Vein ex vivo Assays. Chem. Biol. Interact. 2023, 386, 110746.
[3] Batista, P. R.; de Alencar Silva, A.; de Sena Bastos, C. M.; et al. Vasodilation Promoted by (E,E)-Farnesol Involving Ion Channels in Human Umbilical Arteries. Heliyon 2023, 9, e16985.
[4] Z. Yu, F. Lin, D. Tang, et al., An Insecticidal Composition Containing Farnesol, China Patent CN202210867209.2, 2022.
[5] G. Liu, The Mixture of Farnesol and Nicotine Shows Significant Synergistic Aphicidal Effect, Pesticide Market News 2019, 3:1.
You may like
See also
Lastest Price from (E,E)-Farnesol manufacturers

US $0.00/KG2025-08-30
- CAS:
- 106-28-5
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 200KG

US $0.00-0.00/kg2025-07-04
- CAS:
- 106-28-5
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20MT