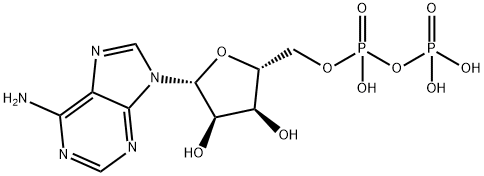
Biosynthesis and Biological Activity of ADP
ADP is a nucleotide composed of adenine, ribose, and two phosphate groups linked by a pyrophosphate bond. As a core molecule in biological energy metabolism, ADP stores energy through phosphorylation to form adenosine triphosphate (ATP) and releases energy for cellular activities upon ATP hydrolysis. This compound appears as a white crystalline powder under ambient conditions, is readily soluble in water, and must be stored sealed under argon at –18°C or below to maintain its stability. In the process of platelet aggregation, ADP-H acts as a key agonist, activating specific receptors to induce hemostatic responses.
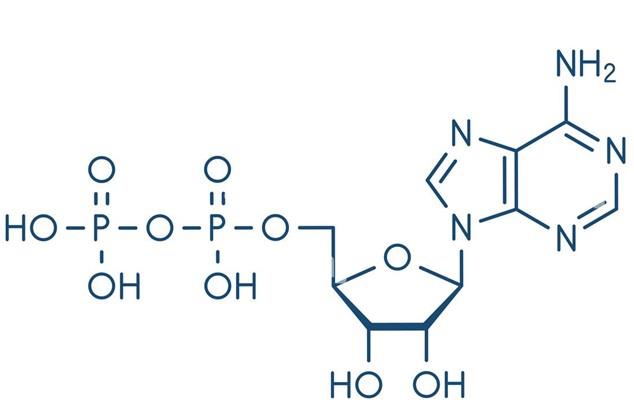
Figure1: Chemical Structure of ADP
Structural Features
ADP is a nucleotide formed by the linkage of adenosine and two phosphate groups. As a crucial organic compound in metabolism, it plays a vital role in energy flow within living cells. A molecule of ADP consists of three key structural components: a sugar backbone attached to an adenine molecule, and two phosphate groups bonded to the 5′ carbon atom of the ribose. The ADP-ATP cycle supplies the energy required to perform work in biological systems, representing a thermodynamic process of energy transfer from one source to another. When ADP combines with a phosphate group and gains 8 kcal/mol of energy, it is converted into ATP. Under normal physiological conditions, discoid platelets circulate freely in the blood without interacting with each other. ADP is stored in dense granules within platelets and released upon platelet activation. ADP interacts with a family of ADP receptors (P2Y1, P2Y12, and P2X1) found on platelets, thereby leading to platelet activation. [1]
Biosynthesis
Within the cell, ADP is primarily generated through the following pathways: ATP releases one phosphate group via hydrolysis to form ADP, a process accompanied by energy release. Adenylate kinase catalyzes the phosphorylation of adenosine monophosphate (AMP) to produce ADP. ADP can be reconverted into ATP through oxidative phosphorylation, substrate-level phosphorylation, and other mechanisms, thereby completing the energy cycle. Additionally, ribonucleotide reductase utilizes ADP-H as a substrate to participate in the synthesis of deoxyribonucleotides.
Biological Activity
The bypdoducts of adenosine 5' -triphosphate (ATP) hydrolysis, ADP and inorganic phosphate (Pi) are released during the cross bridge cycle and can be implicated in the fatigue process due to the requirement of their release for proper crossbridge activity. Pi release is coupled to the powerstroke of the crossbridge cycle. The accumulation of Pi during exercise would lead to a reversal of its release step, therefore causing a decrement in force production capability. Due to the release of Pi with both the immediate (phosphagen) energy system and the hydrolysis of ATP, Pi accumulation is probably the largest contributor to the fatigue process in exercise of any duration. ADP release occurs near the end of the crossbridge cycle and therefore controls the velocity of crossbridge detachment. Therefore, ADP accumulation, which occurs during exercise of extended duration (or in ischaemic conditions), causes a slowing of the rate constants (and therefore a decrease in the maximal velocity of shortening) in the crossbridge cycle and a reduced oscillatory power output. [2]
Animal Experiment
Researchers conducted a study to explore the role of nitric oxide synthesis and release in modulating changes in microvascular permeability of the hamster cheek pouch in response to ADP and bradykinin. Using intravital fluorescence microscopy, the researchers observed changes in the permeability of the hamster cheek pouch to agonists before and after application of nitric oxide enzyme inhibitors—N^G-monomethyl-L-arginine (L-NMMA) and N^W-nitro-L-arginine methyl ester (L-NAME). The increase in permeability was quantified by counting the number of microvascular leaky sites formed. Both ADP and bradykinin significantly increased the number of venular leaky sites, while perfusion with L-NMMA and L-NAME markedly suppressed the increase in microvascular permeability induced by ADP and bradykinin. To verify the specificity of nitric oxide blockade on microvascular permeability, further observations were made on permeability changes induced by adenosine, and the effects of D-NMMA on microvascular permeability were examined. The results showed that L-NMMA treatment did not alter adenosine-induced increases in permeability, and D-NMMA failed to inhibit the increase in microvascular permeability caused by ADP. Therefore, these findings suggest that nitric oxide production, in response to stimulation by ADP and bradykinin, is involved in regulating macromolecular permeability in the hamster cheek pouch in vivo. [3]
Reference
[1] Murugappa S, Kunapuli SP. The role of ADP receptors in platelet function. Front. Biosci. 2006, 11: 1977.
[2] McLester Jr J R. Muscle contraction and fatigue: the role of adenosine 5′-diphosphate and inorganic phosphate[J]. Sports Medicine, 1997, 23(5): 287-305.
[3] Mayhan W G. Role of nitric oxide in modulating permeability of hamster cheek pouch in response to adenosine 5′-diphosphate and bradykinin[J]. Inflammation, 1992, 16: 295-305.
You may like
See also
Lastest Price from ADP manufacturers

US $1.00/kg2025-04-21
- CAS:
- 58-64-0
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 10 mt

US $0.00-0.00/kg2025-04-21
- CAS:
- 58-64-0
- Min. Order:
- 1kg
- Purity:
- ≥95.0%
- Supply Ability:
- 20MT