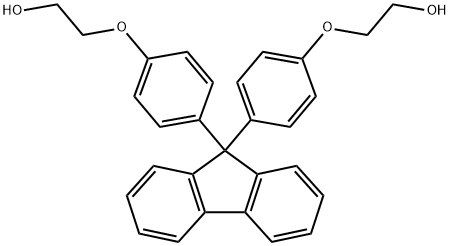
Bisphenoxyethanolfluorene: Monomer & Toxicity
Bisphenoxyethanolfluorene is an aromatic diol organic compound featuring a fluorene ring structure. Its molecular framework comprises a rigid fluorene ring core flanked by phenoxyethyl side chains. This distinctive architecture confers exceptional thermal stability, optical properties, and reactivity. The fluorene ring provides rigidity and high refractive index, while the hydroxyl groups on the side chains can participate in polycondensation reactions such as esterification and etherification. As a key functional monomer, it is primarily used in the synthesis of high-performance resins including epoxies, polycarbonates, and polyesters. Downstream applications are extensive across optical lenses, LCD polarising films, electronic encapsulation materials, OLED displays, and dental materials.

Bisphenoxyethanolfluorene on female development in CD-1 mice
The chemical of concern is Bisphenoxyethanolfluorene (BPEF), which has been listed in the U.S. Environmental Protection Agency (EPA) Distributed Structure-Searchable Toxicity (DSSTox) Database and the European Chemicals Agency (ECHA) Classification and Labeling (C&L) Inventory, however, no toxicological information is available regarding this chemical to date. BPEF has been widely and increasingly used as a monomer or an additive to synthesize polymers in optical products since 1990 s (Fuji et al., 1996). To date, highly functionalized polymers derived from Bisphenoxyethanolfluorene, such as dimethacrylate resins, polyester, cellulose ester, epoxy acrylate, and polycarbonate resin, process superior optical, thermal and mechanical properties. Currently, materials containing BPEFs are widely used in elastomers, coatings, sealants, adhesives photographic film base, polarizer protection film, liquid crystal display industry, digital camera lenses, and dental composite materials. However, there is little information available regarding the risk of BPEF, except that it is classified and labelled as H315 and H319 by the Globally Harmonized System. Thus, this study aimed to test the endocrine disrupting activity of BPEF and investigate its effects on female development in mice, given that its structural analog, fluorene-9-bisphenol (BHPF), is a strong antiestrogenic chemical, which is reported to possess adverse effects on female development and cause adverse pregnancy outcomes in animals. We believe that this study will help fill the toxicological gap of Bisphenoxyethanolfluorene and raise the concerns about BPEF's safety.[1]
In the past few decades, the types and quantities of polymers produced and used by humans have increased dramatically. Various available chemicals are used to produce polymers or released from polymers during their usage. Among them, only a few hazardous chemicals have been identified and assessed for their environmental risks. Here, we reported the antiestrogenic activity of Bisphenoxyethanolfluorene by in vitro, in vivo and in silico approaches. The antiestrogenic activity of BPEF in vitro is so strong which is similar to that of tamoxifen, the antiestrogenic drug. BPEF inhibited the uterine development and caused endometrial atrophies at relatively low doses in adolescent mice after 10-d treatment, and downregulations of BPEF on these estrogen-responsive genes in the uteri of mice might be the underlying mechanisms. BPEF could fit well into the antagonist pocket of human ERα, and the formation of hydrogen bonds and hydrophobic interactions between chemical and the active site of receptor maintained their strong binding. Considering that Bisphenoxyethanolfluorene possesses strong antiestrogenic property and can disrupt female development, we suggest that it should not be used in products that are in close contact with people, particularly immature women. Because no exposure levels have been reported for BPEF so far, we believe that our data indicate a compelling need for investigation of BPEF levels in the environment and humans.
Polycarbonates containing a bisphenol fluorene moiety
Bisphenol A polycarbonate (BPA-PC) is distinguished by its high strength, toughness, and optical clarity, rendering it indispensable in a multitude of optical applications, including lenses, glasses, and display screens. In the wake of rising interest in polymer-based ultra-thin optical lenses and smartphone lenses in recent years, a pivotal area of research has been the enhancement of the polymer's refractive index while concurrently minimizing birefringence. Du et al. copolymerized Bisphenoxyethanolfluorene with BPA, confirming that the refractive index of the copolymer PC could vary between 1.586 and 1.639, and adjusting the copolymer ratio effectively reduced birefringence. Kato et al. synthesized high molecular weight and high refractive index BPEF-PC. However, it still fails to meet the requirements for a high refractive index. To further increase the refractive index, Li et al. introduced BNE and Bisphenoxyethanolfluorene for copolymerization, synthesizing a polycarbonate with a refractive index of up to 1.660. This study employed the melt transesterification method to copolymerize BPEF with BPA and BNE respectively to synthesize PC copolymers. It meticulously evaluates the influence of monomer types and copolymer ratios on the products' Tg, refractive index, and water absorption rate, elucidates the factors affecting hydrolysis and physical aging in hygrothermal environments, and investigates the correlation between hygrothermal aging and the mechanical properties of copolymer PCs.[2]
This study, by copolymerizing BNE with Bisphenoxyethanolfluorene and adjusting the copolymer ratio of BNE to BPEF to 70/30, not only increase the refractive index to 1.659 but also achieve a birefringence value close to zero. The investigation reveals that the incorporation of dihydroxy monomers with elevated pKa values for copolymerization significantly enhanced the hydrolytic stability during hygrothermal aging tests compared to BPA-PC. This suggests that the augmentation of the monomers' pKa value serves as an effective strategy to suppress hydrolysis. Furthermore, the research identify that copolymerized polycarbonates undergo physical aging in an 80 °C hygrothermal environment, with the physical aging window of BNE/BPEF-PC delineated roughly from Tg-45 °C to Tg. The introduction of water causes a reduction in Tg, yet the enthalpy reduction across both dry and hygrothermal conditions can be standardized to a linear relationship by the differential between the aging temperature and Tg. This delineates a strong correlation between the extent of physical aging and Tg. The transparency test shows that the optical transparency of Bis(2-hydroxyethoxy)-1,1′-binaphthyl / Bisphenoxyethanolfluorene -PC shows great stability before and after hygrothermal aging.
Viscoelastic properties of Bisphenoxyethanolfluorene -based cardo polymers
Viscoelastic properties of urethane and ester conjugation cardo polymers that contain fluorene group, Bisphenoxyethanolfluorene (BPEF), were investigated. As for the urethane-type cardo polymers containing BPEF in the main chain, it had a high glass-transition temperature (Tg), which was observed as the α dispersion on viscoelastic measurement, and its temperature depended on the chemical structure of the spacing unit, such as toluene diisocyanate (TDI), 4,4′-methylene diphenyl diisocyanate (MDI), methylene dicycloexyl diisocyanate (CMDI), and hexamethylene diisocyanate (HDI). Moreover, the Tg of urethane-type cardo copolymers with various cardo contents increased with an increase of cardo content. The peak temperature of the αsub dispersion was influenced by the chemical structure of the spacing unit, but it did not change for the cardo polymer containing the same spacing unit. Consequently, it was deduced that the αsub dispersion was originated in the subsegmental molecular motions of the cardo polymers. Ester-type cardo polymer had higher Tg in comparison with noncardo polymer that consisted of dimethyl groups (BPEP) instead of Bisphenoxyethanolfluorene as well. The αsub dispersion was also measured at the temperature between the α and β dispersion, which was assigned to the molecular motion of ester conjugation unit, around 220 K. For ester cardo polymer, the γ dispersion was measured in a low-temperature region around 140 K, and it was due to a small unit motion in the ester-type cardo polymers, such as ethoxyl unit, C2H4O. Moreover, the intensity of the γ dispersion of noncardo polymer was higher than that of cardo polymer, which means the molecular motion was much restricted by the cardo structure of Bisphenoxyethanolfluorene.[3]
References
[1]Jia, Xiaojing et al. “Antiestrogenic property of 9,9-bis[4-(2-hydroxyethoxy)phenyl]fluorene (BPEF) and its effects on female development in CD-1 mice.” Ecotoxicology and environmental safety vol. 242 (2022): 113906. doi:10.1016/j.ecoenv.2022.113906
[2]Lu T, Fang W, Zhou Q, Liu M, Wu G. Synthesis and hygrothermal aging of polycarbonates containing a bisphenol fluorene moiety. RSC Adv. 2024 Jun 3;14(25):17771-17779. doi: 10.1039/d4ra03378g. PMID: 38832245; PMCID: PMC11145624.
[3]Tokumitsu, K., Tanaka, A., Kobori, K., Kozono, Y., Yamada, M., & Nitta, K.-H. (2005). Viscoelastic properties of bis(phenyl)fluorene-based cardo polymers with different chemical structure. Journal of Polymer Science Part B: Polymer Physics, 43(17), 2259–2268.
You may like
Related articles And Qustion
See also
Lastest Price from Bisphenoxyethanolfluorene manufacturers
US $0.00/kg2026-03-20
- CAS:
- 117344-32-8
- Min. Order:
- 1kg
- Purity:
- 99.5%
- Supply Ability:
- 20tons
US $0.00/kg2025-12-10
- CAS:
- 117344-32-8
- Min. Order:
- 25kg
- Purity:
- ≥99.5%
- Supply Ability:
- ton