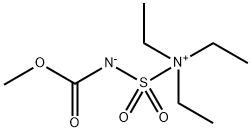
Burgess Reagent: Versatile Dehydrating & Oxidizing Agent
The Burgess reagent is a commercially available compound that has historically found utility as a dehydrating agent. The reagent may also be used to oxidize primary and secondary alcohols to their corresponding aldehydes and ketones in dimethyl sulfoxide. These oxidations were found to be rapid under mild conditions. The methodology is convenient and broadly useful and the oxidation reaction does not evolve toxic byproducts. In the presence of dimethyl sulfoxide, the Burgess reagent efficiently and rapidly facilitates the oxidation of a broad range of primary and secondary alcohols to their corresponding aldehydes and ketones in excellent yields and under mild conditions.

Burgess Reagent Facilitated Alcohol Oxidations in DMSO
In 1968, Atkins and Burgess reported the synthesis of the inner salt [methoxycarbonylsulfamoyl]triethylammonium hydroxide and described its utility in facilitating alcohol dehydrations. Subsequently, the aforementioned compound, which is now colloquially referred to as the “Burgess reagent”, has been successfully employed as a selective and mild dehydrating agent for a broad range of secondary and tertiary alcohols as well as a reagent for converting primary alcohols, primary amides, formamides, and primary nitroalkanes into their respective urethanes, nitriles, isocyanides, and nitrile oxides. The compound has also been employed in cyclodehydrations, which have been particularly useful in peptide modification chemistry, and for the preparation of sulfamides and cyclic sulfamidates from their corresponding amines and epoxides. At nearly the same time that the Burgess reagent was introduced, Parikh and Doering reported (1967) that the sulfur trioxide pyridine complex (C5H5N·SO3) activates dimethyl sulfoxide (DMSO) in the presence of a base (e.g., Et3N) and facilitates the oxidation of primary and secondary alcohols. While the Parikh–Doering oxidation reaction operates under mild conditions, relatively large excesses (3–6 equiv) of pure C5H5N·SO3 are required and, as a result, other activated DMSO-mediated oxidations are often favored. We hypothesized that [methoxycarbonylsulfamoyl]triethylammonium hydroxide, which upon the liberation of triethylamine, should generate an electrophilic dioxosulfimidinum species that may activate DMSO and facilitate oxidation reactions. Herein we report that the Burgess reagent may be used to oxidize primary and secondary alcohols to their respective aldehydes and ketones in the presence of DMSO.[1]
Based on the aforementioned results, we concluded that 1.3 equiv of the Burgess reagent with respect to the alcohol substrate was sufficient and practical. As summarized, a broad range of primary and secondary alcohols were successfully oxidized and their corresponding aldehydes and ketone products in high isolated yields. Over-oxidation of the aldehydes to the corresponding carboxylic acids was not observed. Likewise, olefinic products were not detected when secondary alcohols containing α-hydrogens were subjected to the reaction conditions. In conclusion, we have discovered that the Burgess reagent, a compound that has historically found utility as a dehydrating agent, may be used to oxidize primary and secondary alcohols to their corresponding aldehydes and ketones in dimethyl sulfoxide. The reactions were found to be rapid, operated under mild conditions, and afforded oxidized products in excellent isolated yields. The methodology is advantageous to others in that it is convenient and broadly useful, the oxidation reaction does not evolve toxic byproducts (e.g., carbon monoxide), low temperatures or timed additions are not required, and the oxidation agent is commercially available.
Access to Unprecedented 5-Sulfamido Oxazoles Using Burgess-type Reagents
Oxazoles are heterocycles extensively used by medicinal chemists not only as pharmacophoric groups per se but also as structural elements that impose a degree of conformational restriction. The most common methodology available for the preparation of 5-aminooxazoles bearing a secondary nitrogen at the 5-position is represented by the trifluoroacetic acid (TFA)/trifluoroacetic anhydride (TFAA)-mediated cyclodehydration of diamide/dipeptide precursors. None of the aforementioned cyclodehydrations allows the insertion of a sulfamide moiety in the 5-position of the oxazole ring. Herein we disclose a reaction in which the use of Burgess reagents leads to the simultaneous formation of the oxazole ring and insertion of a sulfamide group on the heterocyclic system. Taking inspiration from the versatile applications of the Burgess reagent, we decided to investigate its dual nature as both a dehydrative and a nondehydrative reagent in a single modality, and we speculated that the application of this reagent to a diamide substructure could result in the formation of oxazoles bearing an N,N′-unsimmetrical sulfamide at position 5.[2]
During the optimization process, it was clear that neither a higher temperature nor a prolonged reaction time favors the formation of the product. In particular, after 1 h, the starting material has usually reacted completely, with the exception of those reactions in which 1 equiv of Burgess reagent is used. In summary, the Ugi reaction mediated by tritylamine as a convenient ammonia surrogate leads to diamide products that, through cyclodehydration triggered by Burgess reagents, are transformed into unprecedented oxazoles bearing N,N′-disubstituted sulfamides at the 5-position. When displaying the benzyl group, the obtained products undergo hydrogenolysis, yielding monosubstitued 5-sulfamido oxazoles. Overall, the sequential synthesis proceeds smoothly and cleanly under mild reaction conditions, is scalable up to 1 mmol, provides high yields, and displays good tolerance to a variety of functional groups, coming to fill a gap in the preparation of both Ugi products and oxazoles.
Synthesis of oxabicyclo[3.2.1]octan-3-ol scaffold via Burgess reagent
Methyl N-(triethylammoniumsulfonyl)carbamate, or better known as the Burgess reagent, was developed by Edward Meredith Burgess that proved to be a powerful selective dehydrating reagent, converting secondary and tertiary alcohols into their corresponding olefins.1 The reaction proceeds via activation of a hydroxyl group to a sulfamate ester as the leaving group, followed by stereospecific syn-elimination according to Zaitsev’s rule. Primary alcohols tend to react differently with Burgess reagent when the intramolecular SN2 pathway is energetically more favorable than the syn-elimination route, thereby converting the primary alcohol into the corresponding carbamate that provides good access to the parent amine upon hydrolysis. This reagent mediated dehydration reactions under mild, neutral conditions enables substrate scope expansion and the application of it as a dehydrating agent. For example, dehydration of oximes and primary amides afford the corresponding nitriles, while dehydration of formamides provide the corresponding isocyanides. Nitrile oxides and carbodiimides are obtained from the corresponding primary nitroalkanes and ureas, respectively, upon treatment with Burgess reagent.[3]
Cyclodehydration of β-hydroxyamides and β-hydroxythioamides using Burgess reagent facilitated access to oxazolines and thiazolines via activation of the hydroxyl group to a sulfamate ester as the leaving group, followed by intramolecular SN2 displacement by the oxygen or sulfur of the amide or the thioamide, respectively. Dehydrative cyclization of δ-diols was achieved using the Burgess reagent under mild conditions to furnish a novel oxabicyclo[3.2.1]octan-3-ol scaffold. The stereochemistry of the generated oxabicyclic compounds was assigned based on single X-ray crystallography and 2D-NMR experiments.
References
[1]Sultane, Prakash R, and Christopher W Bielawski. “Burgess Reagent Facilitated Alcohol Oxidations in DMSO.” The Journal of organic chemistry vol. 82,2 (2017): 1046-1052. doi:10.1021/acs.joc.6b02629
[2]Bhela IP, Serafini M, Del Grosso E, Tron GC, Pirali T. Tritylamine as an Ammonia Surrogate in the Ugi Reaction Provides Access to Unprecedented 5-Sulfamido Oxazoles Using Burgess-type Reagents. Org Lett. 2021 May 7;23(9):3610-3614. doi: 10.1021/acs.orglett.1c01002. Epub 2021 Apr 29. PMID: 33913716; PMCID: PMC8289289.
[3]Elmansy MF, Dos Remedios JRD, Silverman RB. Synthesis of Oxabicyclo[3.2.1]octan-3-ol Scaffold via Burgess Reagent Mediated Cyclodehydration of δ-Diols under Acidic Conditions. Org Lett. 2025 Jan 17;27(2):640-644. doi: 10.1021/acs.orglett.4c04473. Epub 2025 Jan 6. PMID: 39761354; PMCID: PMC11901898.
You may like
See also
Lastest Price from Burgess reagent manufacturers
US $5.00-0.50/KG2024-03-25
- CAS:
- 29684-56-8
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- g-kg-tons, free sample is available

US $15.00-10.00/KG2021-07-02
- CAS:
- 29684-56-8
- Min. Order:
- 1KG
- Purity:
- 99%+ HPLC
- Supply Ability:
- Monthly supply of 1 ton