Catalyst development for hydrogen storage in N-ethylcarbazole
Introduction
Liquid Organic Hydrogen Carriers (LOHCs) hydrogen storage has the advantages of high hydrogen storage density, good safety, high reversibility and convenient transportation, which makes it a promising hydrogen storage technology for application. N-ethylcarbazole (NEC), which has a hydrogen storage capacity of 5.79 wt% and a unique nitrogen heterocyclic ring in its structure, significantly reduces the enthalpy of dehydrogenation and the temperature of the dehydrogenation reaction and facilitates the release of H2, and is considered as one of the ideal LOHCs. [1] As a new liquid organic hydrogen storage carrier, N-ethylcarbazole can be completely hydrogenated to perhydro N-ethylarbazole, which can be stored and transported in the same way as common oil products used. Because of the high mass density and the lower dehydrogenation temperature compared with the traditional materials, N-ethylcarbazole has been considered as one of the most promising hydrogen carriers.[2]
N-ethylcarbazole as a carrier in the LOHC system
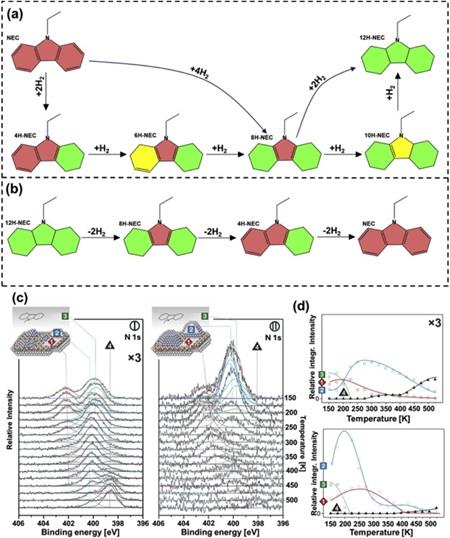
Structurally, N-ethylcarbazole is obtained by adding an ethyl group to carbazole, which reduces the melting point from 243 °C for carbazole to 68 °C for NEC. The heteroatom in N-ethylcarbazole reduces the dehydrogenation enthalpy change, making N-ethylcarbazole competitive against other LOHCs. However, the current significant challenges encountered in NEC/12H-NEC include the production of undesirable by-products due to easy thermal alteration of the N-ethyl group and the high H2 pressure required for reverse hydrogenation (68 bar). To effectively implement N-ethylcarbazole as a LOHC, it is essential to know the mechanism of hydrogen storage in N-ethylcarbazole.The detailed hydrogenation and dehydrogenation processes are shown in Fig. 1(a–b). The hydrogenation enthalpy of N-ethylcarbazole is −50.6 kJ/mol-H2, which means the reaction is exothermic. During dehydrogenation, as seen in Fig. 1(b), 8H-NEC is formed due to the preferable removal of hydrogen from the middle ring. In the next step, it either directly forms 4H-NEC, detaching four hydrogen atoms from a side carbon ring, or first forms 6H-NEC and then releases the two hydrogen atoms to form 4H-NEC. In the end, hydrogen-depleted NEC is formed after removing four hydrogen atoms from the other side of the ring. The fully hydrogenated N-ethylcarbazole (12H-NEC), partially hydrogenated intermediates like octahydro-NEC (8H-NEC) and tetrahydro-NEC (4H-NEC)) are in the liquid state at room temperature, while N-ethylcarbazole is solid below 68 °C.
To further understand the dehydrogenation process, it is crucial to understand the interaction between reactant and catalyst. Amende et al. showed that at temperatures up to −50 °C, molecular adsorption of 12H-N-ethylcarbazole takes place on Pt(111), without molecular decomposition below −100 °C. They also showed that at cryogenic temperatures, 12H-NEC adsorbs molecularly on Pd nanoparticles and Pd(111) . The onset temperature of dealkylation on defect-rich Pt aggregates is about 90 K lower than on single crystal Pt(111) , as shown in Fig. 1(c–d). The dealkylation is attributed to easier N-ethyl bond breakage triggered by low-coordination Pt sites, whose population increases as the size of Pt particles decreases. Dehydrogenation starts by activating the C–H bond directly near the nitrogen atom in 12H-N-ethylcarbazole, resulting in 8H-NEC being the first stable intermediate product. The transition from 12H-NEC to 8H-NEC occurs at temperatures above −50 °C, increasing in rate above 0 °C. Their findings align with computations showing 8H-N-ethylcarbazole to be the first intermediate dehydrogenation product, a result that we have independently confirmed.[3]
Catalysts for N-ethylcarbazole
Efficient catalysts are crucial for successfully implementing LOHC technology and should have the ability to achieve >80% hydrogenation/dehydrogenation of the hydrogen carrier under the following conditions (Temperature: <200 °C, pressure: <10 MPa). Also, a stable catalyst during the hydrogen storage cycle can have a significant economic impact. If the catalyst changes in phase during the hydrogen storage cycle, the new phase formed should be favourable for the process. During its functioning, catalysts are expected to have optimum binding to the reaction intermediates. In the case of weak binding, the reactant's adsorption to the substrate is complex, resulting in low catalytic activity. On the other hand, strong binding limits the regeneration of catalytic surface sites. The binding depends on many factors, such as crystal structure, material, morphology, and particle size. For LOHC technology, the design of catalysts can be mainly focussed on accelerating low-temperature dehydrogenation to overcome the thermodynamic barrier, in view that compared to hydrogenation, endothermic dehydrogenation requires high heat . For efficient large-scale N-ethylcarbazole application, nano catalysts can be prepared through top-down approaches, such as ball milling, and bottom-up approaches, such as hydrothermal synthesis. However, it should be noted that before proceeding to large-scale applications, it is essential to test the hydrogenation reaction on a pilot scale using catalyst modules such as a fluidized bed since it is not practical to use suspended powder catalysts in large-scale applications. A combination of techno-economic analysis (TEA) and life cycle assessment (LCA) should be conducted to understand the process of hydrogenation of N-ethylcarbazole better before proceeding towards large-scale application. In the following sections, we discuss several vital catalysts for N-ethylcarbazole studied to date, including ruthenium-, nickel-, palladium-, and platinum-based catalysts.[3]
References
[1]Liu Ty.Study on Hydrogen Storage Performance of N-Ethylcarbazole Catalyzed by SBA-15-Supported Ruthenium-Based Catalysts[D].Heilongjiang University,2024.DOI:10.27123/d.cnki.ghlju.2024.000104.
[2] Chen B. Study on the hydrogen storage performance of N-ethylcarbazoleover Ni-based catalysts[D].Tianjin University of Science and Technology,2022.DOI:10.27359/d.cnki.gtqgu.2022.000988.
[3]Permude P , Ahmad A , Chen H ,et al.Effective catalysts for typical liquid organic hydrogen carrier N-Ethylcarbazole[J].International journal of hydrogen energy, 2025(Jan.):98.DOI:10.1016/j.ijhydene.2024.12.038.
Related articles And Qustion
See also
Lastest Price from N-Ethylcarbazole manufacturers
US $1.10-9.90/kg2025-08-15
- CAS:
- 86-28-2
- Min. Order:
- 100kg
- Purity:
- 99%min
- Supply Ability:
- 100kg

US $0.00/KG2025-04-21
- CAS:
- 86-28-2
- Min. Order:
- 1KG
- Purity:
- 98%min
- Supply Ability:
- 30tons/month