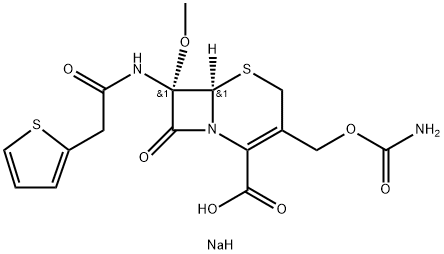
Cefoxitin Sodium: Antibiotic Profile & Clinical Applications
Cefoxitin sodium is an intravenous (IV) antibiotic used to treat certain infections caused by bacteria. It works by killing the bacteria that cause the infection. Common side effects include diarrhea or pain, itching, swelling, or redness near the injection site. Serious side effects are much less common, but can include a type of diarrhea that is difficult to treat. Notify your healthcare provider right away if you develop watery or bloody diarrhea. Cefoxitin sodium may not be appropriate for some of the infections listed above, depending on which germs are causing the infection. Bacteria in some locations may have developed resistance to cefoxitin, which will make it not work as well.

Cefoxitin sodium Does Not Induce Production of Penicillin Binding Protein 2a
Beta-lactam resistance in methicillin-resistant staphylococci (MRS) is caused by the expression of penicillin-binding protein 2a (PBP2a), encoded by the mecA gene, which has low binding affinities to practically all beta-lactam antibiotics so far introduced into clinical use. Detection of this resistance is complicated because different methicillin resistance levels are expressed in the staphylococcus population. Recently the cefoxitin disk has been proposed as an alternative method of detecting MRS. Cefoxitin sodium is a cephamycin-type antibiotic, and it has been described as an inducer of methicillin resistance by production of the PBP2a. The paper published by Annie Felten et al. has been used as a reference suggesting the use of the cefoxitin disk as being helpful in oxacillin resistance detection. According to these authors, “surprisingly, cefoxitin sodium induced the PBP2a production in vitro in MSSA isolates for which cefoxitin MICs were high.” The methicillin-susceptible Staphylococcus aureus (MSSA) strain referred to by these authors is S. aureus N315, which was isolated in Japan in 1982, and at that time it was identified as methicillin susceptible and cefoxitin sodium resistant. This strain became resistant to methicillin after induction by cefoxitin. In 1992, it was known that this strain would not be classified as MSSA because it is a pre-methicillin-resistant S. aureus (MRSA) strain.[1]
When many researchers and clinical bacteriologists are looking for alternative methods for detection of low-level methicillin resistance, it is very important to explain that cefoxitin sodium does not induce PBP2a production in vitro in MSSA isolates unless this isolate is pre-MRSA, with a functional mecI gene. Based on published data, it may be postulated that low-level methicillin resistance in staphylococcus strains could be easily detected if the isolate was also resistant to cefoxitin sodium due to the participation of native PBP2. We did not exclude the possibility of the mechanism's association with participation of several native PBPs that permit the expression of methicillin resistance. The mechanism of resistance to β-lactam in staphylococci is very complex, involving the participation of acquired and native genes. The function of each gene that participates in this mechanism of resistance is not completely understood, and this represents a challenge for researchers.
cefoxitin sodium, a Semisynthetic Cephamycin Antibiotic
The in vitro activity of cefoxitin sodium, 3-carbamolyloxymethyl-7-α-methoxy-7[2-(2-thienyl)acetamido]-3-cephem-4-carboyxlic acid, was investigated. Activity against gram-positive organisms was less than that of cephalothin and cephloridine. It was highly active against gram-negative bacilli, with activity against Escherichia coli, Proteus mirabilis, and Klebsiella pneumoniae equal to that of currently available cephalosporins. In addition, it was active against certain Enterobacter strains, Serratia marcescens, indole-positive Proteae and Herellea. The strains of these latter bacteria were strains susceptible to carbenicillin and ticarcillin. Pseudomonas aeruginosa and other Pseudomonas species were resistant. Changes in pH, inoculum size, and type of growth medium had no significant effect on the activity of the antibiotic. Cefoxitin sodium was highly resistant to hydrolysis by various types of gram-negative beta-lactamases. The precise role of resistance to beta-lactamase hydrolysis varied from strain to strain. Bacterial resistance to cefoxitin was not necessarily related to hydrolysis of the antibiotic. However, the resistance of cefoxitin to hydrolysis did contribute to its activity. Cefoxitin sodium could function as an inducer of beta-lactamase activity and effectively bound to purified beta-lactamases.[2]
Cefoxitin sodium as definitive treatment for Klebsiella pneumoniae bacteremia
Since the 1990s, the emergence and diffusion of extended-spectrum beta-lactamase-producing Enterobacterales (ESBL-E) has been a global concern. In 2021, the rate of invasive ESBL-E isolates has reached 7.5% in French healthcare settings. ESBL-Klebsiella pneumoniae (ESBL-KP) infections are also common nosocomial infection, but clinical data on their treatment with cefoxitin sodium are scarce. In Guadeloupe University Hospital, the observed incidence rate of nosocomial ESBL-E infections is one of the highest in France, with a large predominance of Klebsiella pneumoniae. Since 2015, to limit the use of carbapenems, antibiotic guidelines in our intensive care unit (ICU) proposed cefoxitin sodium rather than carbapenems as definitive treatment in cefoxitin-susceptible ESBL-E systemic infections, whenever clinical condition and microbiological findings are compatible. Until now this therapeutic strategy had not been formally evaluated. The aim of our study was thus to compare the clinical efficacy of cefoxitin as definitive antibiotic therapy for ICU patients with ESBL-KP bacteremia, versus standard carbapenem therapy. Selection of cefoxitin- and carbapenem-resistant Enterobacterales in both groups was also analyzed.[3]
A total of 110 patients with bloodstream infections were enrolled. Sixty-three patients (57%) received definitive antibiotic therapy with cefoxitin, while forty-seven (43%) were treated with carbapenems. 30-day clinical success was not significantly different between patients treated with cefoxitin sodium (57%) and carbapenems (53%, p = 0.823). PS-adjusted and PS-matched analysis confirmed these findings. Change of definitive antibiotic therapy was more frequent in the cefoxitin group (17% vs. 0%, p = 0.002). No significant differences were observed for the other secondary endpoints. The acquisition of carbapenem-resistant Pseudomonas aeruginosa was significantly higher in patients receiving carbapenem therapy (5% vs. 23%, p = 0.007).
References
[1]da Costa Darini AL, Palazzo IC. Cefoxitin does not induce production of penicillin binding protein 2a in methicillin-susceptible Staphylococcus aureus strains. J Clin Microbiol. 2004 Sep;42(9):4412; author reply 4412-3. doi: 10.1128/JCM.42.9.4412-4413.2004. PMID: 15365061; PMCID: PMC516335.
[2]Neu HC. Cefoxitin, a semisynthetic cephamycin antibiotic: antibacterial spectrum and resistance to hydrolysis by gram-negative beta-lactamases. Antimicrob Agents Chemother. 1974 Aug;6(2):170-6. doi: 10.1128/AAC.6.2.170. PMID: 15828188; PMCID: PMC444623.
[3]Dequidt T, Bastian S, Nacher M, Breurec S, Carles M, Thiery G, Camous L, Tressieres B, Valette M, Pommier JD. Cefoxitin versus carbapenems as definitive treatment for extended-spectrum β-lactamase-producing Klebsiella pneumoniae bacteremia in intensive care unit: a propensity-matched retrospective analysis. Crit Care. 2023 Nov 1;27(1):418. doi: 10.1186/s13054-023-04712-2. PMID: 37915017; PMCID: PMC10619259.
You may like
Related articles And Qustion
Lastest Price from Cefoxitin sodium manufacturers

US $0.00/KG/Tin2025-04-21
- CAS:
- 33564-30-6
- Min. Order:
- 10KG
- Purity:
- 927-970μg/mg Sterile USP
- Supply Ability:
- 100kg

US $1.00/KG2025-04-21
- CAS:
- 33564-30-6
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10mt