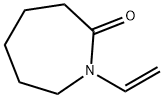
Characteristics and Polymerization Method of N-Vinylcaprolactam
N-Vinylcaprolactam is a vinyl monomer suitable for photocuring reactions and can be used in UV polymerization of various unsaturated systems such as UV coatings, UV inks, and UV adhesives. It holds significant applications in the synthesis of polymer materials. As a monomer compatible with free‑radical curing systems, N-Vinylcaprolactam demonstrates excellent curing speed, adhesion, and dilution performance, particularly in the field of high‑performance UV inks, especially for inkjet printing, making it a mainstream UV raw material.

Figure1: Picture of N-Vinylcaprolactam
Characteristics
The chemical reactivity of N-Vinylcaprolactam is primarily concentrated on the double‑bond unit in its structure, which can undergo polymerization under specific conditions. N-Vinylcaprolactam offers a balanced hardness and toughness, exhibiting outstanding adhesion on a variety of metal substrates. At 50 °C, N-Vinylcaprolactam has a viscosity of 2.88 mPa·s and shows good dilution performance, rendering it highly recommended for UV inkjet and UV screen‑printing inks. Being an organosoluble amphiphilic material, it is soluble in solvents such as benzene, isobutanol, and isopropanol. Additionally, this compound can be polymerized via free‑radical polymerization at temperatures above its melting point to produce poly(NVCL).
Synthesis
The synthesis of N-Vinylcaprolactam (NVCL) was investigated using acetylene as the vinylation agent in three distinct reactor systems: a 500 mL stirred reactor, a 500 mL tower reactor, and an 8,000 mL jet loop reactor. With potassium caprolactam as the catalyst and 18‑crown‑6 as the co‑catalyst, N-Vinylcaprolactam was produced through the reaction of acetylene with caprolactam (CL) under atmospheric pressure. The resulting crude product was purified by vacuum distillation followed by further distillation to obtain high‑purity N-Vinylcaprolactam (98%). The product structure was characterized by FTIR and GC/MS analysis. The CL conversion and product selectivity were 30.4% and 73.4% in the stirred tank reactor, 31.4% and 79.2% in the tower reactor, and 56.7% and 85.6% in the jet loop reactor, respectively. Resinification of the reaction mixture was identified as a key factor in controlling the success of the synthesis. [1]
Polymerization Method
Method 1
Poly(N-Vinylcaprolactam), abbreviated as PNVCL, is a nonionic, nontoxic, water‑soluble, thermally responsive, and biocompatible polymer. It contains hydrophilic carboxylic and amide groups along with a hydrophobic carbon‑carbon backbone, making it suitable for biomedical applications. In this study, N-Vinylcaprolactam was polymerized via free‑radical polymerization at 50, 60, and 70 °C. The resulting polymers were obtained as white powders, soluble in water and common organic solvents. The plot of percent conversion versus time is nearly linear up to approximately 60 % conversion, with no observable induction period. Based on the Arrhenius plot, the activation energy of the polymerization was calculated to be 108.4 kJ/mol. FTIR and NMR analyses confirmed that N-Vinylcaprolactam polymerizes through the opening of the carbon‑carbon double bond, while the caprolactam ring remains unchanged. The polymer was further characterized using FTIR, ¹H‑NMR, ¹³C‑NMR, DSC, TGA, and XRD techniques. [2]
Method 2
Poly(N-Vinylcaprolactam) (PNVCL) is currently one of the most widely used and effective new‑generation kinetic inhibitors for natural gas hydrates, serving as an excellent alternative to conventional thermodynamic inhibitors. Using methanol (AM) as the solvent, azobisisobutyrimidine hydrochloride (AIBI) as the initiator, and N-Vinylcaprolactam (NVCL) as the monomer, PNVCL was synthesized via polymerization. The effects of reaction temperature, time, solvent amount, and other synthesis conditions on the molecular weight (Mn) and yield of the product were investigated to determine an appropriate synthesis process. The results showed that when the feed mass ratio was m(NVCL):m(AIBI):m(AM) = 10:0.03:19.8, the system temperature was 65 °C, and the reaction time was 6 h, the laboratory‑scale yield reached 95.4%. Based on these laboratory conditions, scale‑up experiments were conducted, and the optimal scaled‑up process conditions were determined as follows: m(NVCL):m(AIBI):m(AM) = 10:0.03:21, system temperature 65–68 °C, reaction time 5–6 h, yielding PNVCL with a recovery of 93.4%, demonstrating good scalability. [3]
Reference
[1] Er-Li Z .Synthesis of N-Vinylcaprolactam[J].Fine chemicals, 2008.
[2] Selin Kozano.Polymerization of N-Vinylcaprolactam and Characterization of Poly(N-Vinylcaprolactam)[J].Journal of Macromolecular Science, Part A: Pure and Applied Chemistry, 2011, 48: 467-477.
[3] Xu, S., Geng, L., Pan, B., et al. Synthesis and Scale-up of Poly(N-Vinylcaprolactam) [J]. 2017.
You may like
Lastest Price from N-Vinylcaprolactam manufacturers

US $0.00/kg2025-08-05
- CAS:
- 2235-00-9
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 50 tons

US $9.90/KG2025-04-21
- CAS:
- 2235-00-9
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt