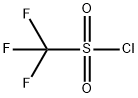
Chemical Properties and Applications of Trifluoromethanesulfonyl chloride
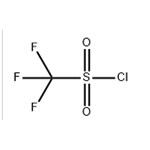
Trifluoromethanesulfonyl chloride appears as a colorless to pale yellow liquid with notable hygroscopic and deliquescent properties. It tends to undergo hydrolysis and degradation when exposed to water or alcohols. Trifluoromethanesulfonyl chloride is primarily used as a trifluoromethylating reagent in ruthenium(II)‑complex‑catalyzed reactions of aromatic compounds and alkenes, yielding downstream products such as N‑phenylbis(trifluoromethanesulfonyl)imide. Synthetic routes to this compound include fluorination of methanesulfonyl chloride, chlorine oxidation of trifluoromethyl thioethers, or reaction with phosphorus trichloride, among other methods.
Chemical Properties
Trifluoromethanesulfonyl chloride (CF₃SO₂Cl) serves as an efficient sulfonating agent for alcohols, and it can also function as a chlorinating agent for carbanions. The rate of chlorination of carbanions is approximately 10⁵ times faster than that of alcohol sulfonation. These properties have been exploited in the synthesis of 2,2‑disubstituted tetrahydropyrans by treating the corresponding 5,5‑disubstituted pentan‑1‑ol with trifluoromethanesulfonyl chloride in the presence of Et₃N and CH₂Cl₂. Researchers have reported further applications of this strategy for constructing heterocycles fused with β‑lactams, including O₃‑isooxacepham and the biologically active isopenam. [1]
Acylation Reaction
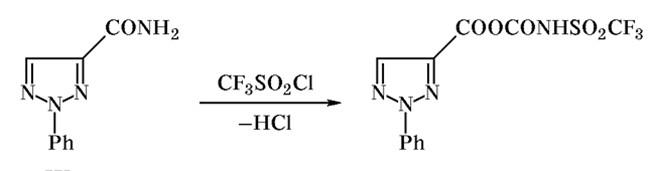
Figure1: Acylation Reaction of Trifluoromethanesulfonyl chloride
To a solution of 1,2,3-triazole sodium salt in tetrahydrofuran we added dropwise under stirring and cooling an equimolar amount of trifluoromethanesulfonyl chloride in THF. The mixture was stirred for 1 h, heated for 2 h under reflux, and cooled. The precipitate was filtered off, and the filtrate was evaporated. The viscous semicrys talline residue underwent spontaneous decomposition with strong heat evolution and tarring on exposure to air (in an attempt to dry the product).[2]
Cyclization Reaction
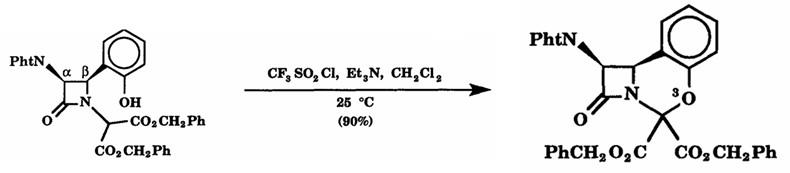
Figure2: Cyclization Reaction of Trifluoromethanesulfonyl chloride
To a solution of 3 (2.95 g, 5.00 retool) in CH2C12 (50 mL) was added Et3N (0.610 g, 6.03 mmol). Trifluoromethanesulfonyl chloride (0.86 g, 5.1 mrnol) in CH2C12 (5.0 mL) was added dropwise to the reaction mixture at 0 °C over a period of 5.0 rain. After the mixture was warmed up to room temperature, it was concentrated under reduced pressure to dryness and then Et20 was added. The ethereal solution was washed with H20, dried over MgSO4 (s), and treated with charcoal. After filtration, evaporation, and purification by use of column chromatography (silica gel, CHC13 as eluant), compound 5 (2.65 g, 4.50 mmol) was isolated in 90% yield as a foam. [1]
Benzyl C-H chlorination
Unlike traditional redox processes, light-induced ligand-to-metal charge transfer (LMCT) has recently emerged as an effective synthetic strategy for generating reactive radical species, which can participate in intermolecular hydrogen atom transfer (HAT) processes to achieve C–H activation. Researchers have investigated the influence of different chlorinating reagents on the reaction regioselectivity using chlorine radicals as HAT reagents, where the chlorine radicals are generated via light-induced LMCT between Fe(III) and chloride ions. It was found that trifluoromethanesulfonyl chloride demonstrates exceptionally high benzylic selectivity toward diverse alkylbenzenes, thereby providing a practical protocol for the synthesis of benzylic chlorides. [3]
Chemical Applications
Trifluoromethanesulfonyl chloride can be employed as a pharmaceutical intermediate, for instance, in the preparation of trimethylresveratrol. Trimethylresveratrol serves as a key synthetic precursor for many antibacterial agents, weight‑loss drugs, anticancer therapeutics, and auxiliary agents. Examples include the synthesis of anticancer‑active compounds such as H‑PGDS inhibitors, Rho‑kinase inhibitors, KSP inhibitors, CSF‑1R inhibitors, and renin inhibitors. Furthermore, trifluoromethanesulfonyl chloride can be prepared by reacting trifluoromethanesulfonic acid with thionyl chloride.
Sulfenylation Reaction
Researchers developed a novel method for the synthesis of trifluoromethyl disulfides and difluoromethyl disulfides through triphenylphosphine‑mediated, metal‑free deoxygenation‑trifluoromethylthiolation and deoxygenation‑difluoromethylthiolation of thiols, using trifluoromethanesulfonyl chloride and CHF₂SO₂Cl as the respective sulfenylation reagents. It was found that iodine generated in situ from iodide promotes the reaction by forming iodotriphenylphosphonium iodide, which acts as a reducing agent in this transformation. The ready availability of reagents, mild reaction conditions, and absence of transition metals make this protocol more practical than traditional approaches. [4]
Reference
[1] Meshcheryakov V I, Shainyan B A, Tolstikova L L, et al. Reactions of 1, 2, 3-triazoles with trifluoromethanesulfonyl chloride and trifluoromethanesulfonic anhydride[J]. Russian journal of organic chemistry, 2003, 39(10): 1517-1521.
[2] Hwu J R, Hakimelahi S, Lu K L, et al. Different roles of trifluoromethanesulfonyl chloride in the construction of heterocycles fused with β-lactams[J]. Tetrahedron, 1999, 55(26): 8039-8044.
[3] Yang C X, Liu D G, Li L J, et al. Light‐induced FeCl3‐catalyzed selective benzyl C? H chlorination with trifluoromethanesulfonyl chloride[J]. Advanced Synthesis & Catalysis, 2023, 365(23): 4144-4149.
[4] Zhao X, Li T, Yang B, et al. Transition-metal-free trifluoromethylthiolation and difluoromethylthiolation of thiols with trifluoromethanesulfonyl chloride and difluoromethanesulfonyl chloride[J]. Tetrahedron, 2017, 73: 3112-3117.
You may like
See also
Lastest Price from Trifluoromethanesulfonyl chloride manufacturers
US $0.00/KG2025-11-21
- CAS:
- 421-83-0
- Min. Order:
- 2000KG
- Purity:
- 99.9%
- Supply Ability:
- 20tons
US $0.00-0.00/kg2025-04-04
- CAS:
- 421-83-0
- Min. Order:
- 1kg
- Purity:
- 98%
- Supply Ability:
- 1Ton