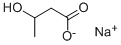Chemical Properties and Preparation Methods of DL-3-Hydroxybutyric acid sodium salt
DL-3-Hydroxybutyric acid sodium salt is an organic acid sodium salt that appears as a white solid powder under normal temperature and pressure. It exhibits significant alkalinity and can undergo acid-base neutralization reactions with common acidic substances. It is sparingly soluble in diethyl ether but soluble in water and alcohol-based organic solvents. DL-3-Hydroxybutyric acid sodium salt is a metabolite that is elevated in type 1 diabetes. It can modulate the properties of membrane lipids and has promising applications in basic biochemistry research.
Chemical Properties
DL-3-Hydroxybutyric acid sodium salt, chemically characterized as a carboxylate salt, exhibits pronounced nucleophilic character that enables its participation in substitution reactions with common electrophilic reagents, such as benzyl bromides or benzyl chlorides, within polar organic solvent systems, thereby facilitating the formation of the corresponding carboxylic ester derivatives through nucleophilic acyl substitution mechanisms.
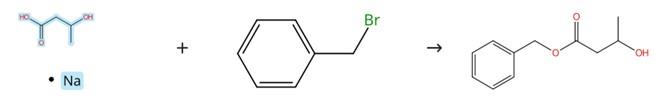
Figure1: Alkylation reaction of DL-3-hydroxybutyric acid sodium salt
In a thoroughly dried reaction flask, DL-3-hydroxybutyric acid sodium salt was dissolved in anhydrous N,N-dimethylformamide under an inert atmosphere, after which benzyl bromide was introduced slowly to the reaction mixture with continuous stirring. The resulting reaction mixture was subsequently heated to 60°C and maintained at this temperature with stirring overnight to ensure complete conversion. Upon completion of the reaction as monitored by appropriate analytical techniques, the reaction mixture was carefully quenched by the addition of aqueous ammonium chloride solution, followed by extraction with ethyl acetate to recover the desired product. The combined organic layers were collected, washed sequentially with water and brine to remove any residual water-soluble impurities, and then dried thoroughly over anhydrous sodium sulfate. After filtration to remove the drying agent, the filtrate was concentrated under reduced pressure using a rotary evaporator to yield a crude residue, which was subsequently purified by silica gel column chromatography employing an appropriate gradient elution system to afford the target benzyl ester derivative. [1]
Biological Activity
DL-3-Hydroxybutyric acid sodium salt interacts with lipids (modeled using DPPC monolayers) and alters phase behavior at clinically relevant concentrations. It also reduces the interfacial viscosity of DPPC monolayers.
Preparation Methods
In a Schlenk tube, combine 1,3-butadiol (5.5 mmol), cyclohexanone (2.8 mL, 27.5 mmol, 5 equiv), water (6.5 mL, 362 mmol, 66 equiv), and sodium hydroxide (263 mg, 6.6 mmol, 1.2 equiv). Add dodecane (0.68 mL, 3 mmol) as an internal standard, and degas the biphasic mixture by purging with argon for 15 minutes. Under a continuous argon flow, introduce Rh(trop2NH)(PPh3)(OTf) (5 mg, 5.5 μmol, 0.1 mol%) and stir the reaction at room temperature for 12 hours. After completion, remove all volatile components (both aqueous and organic) under reduced pressure, then dissolve the residue in water. Wash the aqueous solution twice with small portions of diethyl ether, acidify with 2 M hydrochloric acid, and evaporate the water under reduced pressure. Finally, recrystallize the residue from hot toluene to give DL-3-Hydroxybutyric acid sodium salt. The reaction progress can be monitored by gas chromatography, if desired, by comparing the alcohol product to the dodecane internal standard. [2]
Biological Research
Cell viability was assessed using the Cell Counting Kit-8 (CCK-8) assay. PC12 cells were cultured and treated with CoCl₂, resveratrol (RES), and DL-3-Hydroxybutyric acid sodium salt, either alone or in combination. To evaluate the cytotoxic effects of CoCl₂, cells were exposed to concentrations of 5 µM, 50 µM, or 100 µM in 96-well plates for 24, 48, or 72 hours, with the 48-hour time point selected for subsequent experiments. The potential protective effects of RES (50 µM) and DL-3-Hydroxybutyric acid sodium salt were assessed by co-administering them with CoCl₂. Four hours before the end of each incubation period, 10 µL of CCK-8 solution was added to each well. Cytotoxicity was determined by measuring the absorbance difference at 450 nm. For each experimental group, replicate measurements were averaged, and cell viability was calculated as a percentage relative to untreated control cells, with results presented as means ± standard deviations (SD). [3]
Reference
[1] Pharmaceutical composition containing ketone ester compound for the treatment and prophylaxis of congestive heart failure, United States Patent, Patent number:US20250282707 A1.
[2] Zweifel, Theo; et al, Catalyzed dehydrogenative coupling of primary alcohols with water, methanol, or amines, Angewandte Chemie, International Edition (2009), 48(3), 559-563.
[3] Wang Y, Zhao J, Sun L, et al. Resveratrol attenuates the CoCl2-induced hypoxia damage by regulation of lysine β-hydroxybutyrylation in PC12 cells[J]. BMC neurology, 2025, 25(1): 153.
You may like
Lastest Price from DL-3-Hydroxybutyric acid sodium salt manufacturers

US $0.00-0.00/kg2025-07-28
- CAS:
- 150-83-4
- Min. Order:
- 1kg
- Purity:
- 99%min
- Supply Ability:
- 1000kg

US $10.00/KG2025-04-21
- CAS:
- 150-83-4
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt