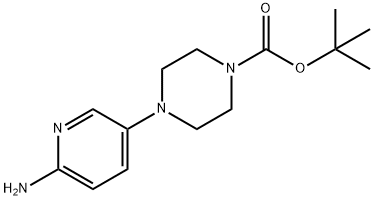
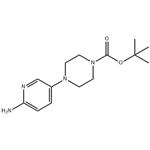
Chemical Property of Tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate
Tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate is a pyridine derivative that exists as a pale yellow to brown solid at room temperature and under normal pressure. It exhibits significant basicity and excellent chemical stability. This compound is insoluble in water but soluble in alcoholic organic solvents and ethyl acetate. Tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate is primarily used as a pharmaceutical chemical intermediate, with studies reporting its application in the synthesis of the drug molecule Palbociclib.
Property
Tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate contains a pyridine unit and multiple amino groups in its structure, exhibiting significant basicity and the ability to undergo acid-base neutralization reactions with most acidic substances. The amino units in its structure also display notable nucleophilicity, enabling condensation reactions with common electrophilic reagents such as acetyl chloride to yield the corresponding N-acylated derivatives.
Preparation Method
Method 1
Tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate was obtained through an SNAr reaction starting with piperazine and 2-nitro-5-halopyridine, followed by N-protection and catalytic hydrogenation.
Method 2
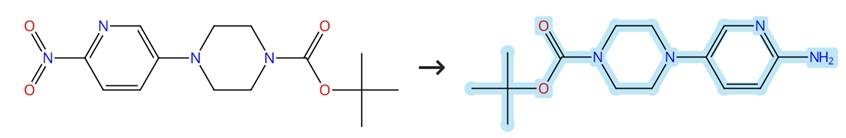
Figure1: Synthesis of Tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate
Tert-butyl 4-(6-nitropyridin-3-yl)piperazine-1-carboxylate (1.0 mmol), iron powder (336 mg, 3.0 mmol), and NH4Cl (481 mg, 9.0 mmol) were dissolved in 70% EtOH, and the reaction mixture was stirred at 70°C under an argon atmosphere overnight. After concentration in vacuo, the residue was purified on a silica gel column to afford tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate. [1]
Aromatic Nucleophilic Substitution Reaction
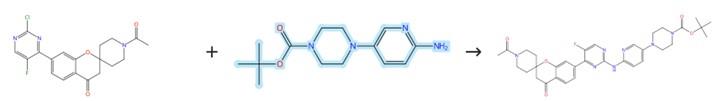
Figure2: Aromatic Nucleophilic Substitution of Tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate
Under a nitrogen atmosphere, tert-butyl 4-(6-aminopyridin-3-yl) piperazine-1-carboxylate (11.02 g, 39.6 mmol) was added to a solution of 6-bromo-8-cyclopentyl-5-methyl-2-(methylsulfinyl)pyrido[2,3-d]pyrimidin-7(8H)-one (10, 9.80 g, 26.4 mmol) in dimethyl carbonate (100 mL). Following the addition, the reaction mixture was stirred at 110 °C for 25 hours under nitrogen protection. After this period, the reaction mixture was cooled to room temperature, diluted with water, and extracted with dichloromethane and saturated NaHCO₃ solution. The combined organic phases were dried over MgSO₄, filtered, and concentrated in vacuo. The residue was purified by passing the organic extract through a silica gel column eluting with DCM/MeOH (40:1) to afford the product. [2]
Coupling Reaction
In a 25 mL sealing tube, Tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate (220 mg, 0.7 mmol), Spiro[2H-1-benzopyran-2,4′-piperidin]-4(3H)-one, 1′-acetyl-7-(2-chloro-5-fluoro-4-pyrimidinyl) (162 mg, 0.84 mmol), Pd2(dba)3 (64 mg, 0.07 mmol), BINAP (87 mg, 0.14 mmol), Cs2CO3 (456 mg, 1.4 mmol) and dioxane (8 mL) were combined, and the resulting mixture was stirred at 100°C for 12 hours under a nitrogen atmosphere. Upon completion, the reaction mixture was allowed to cool to room temperature, after which the solvent was removed by concentration under vacuum, and the residue thus obtained was subsequently purified by silica gel column chromatography. [3]
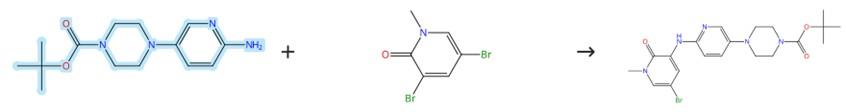
Figure3: Coupling Reaction of Tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate
A 250 mL three-neck round-bottom flask equipped with a magnetic stirrer and reflux condenser was charged with Tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate (2 g, 7.185 mmol), 3,5-dibromo-1-methylpyridin-2(1H)-one (1.74 g, 6.53 mmol), Cs2CO3 (2.98 g, 9.14 mmol), xantphos (302.3 mg, 0.522 mmol), and Pd2(dba)3 (239.2 mg, 0.261 mmol). Under a nitrogen atmosphere, 1,4-dioxane (30 mL) was added, and the mixture was stirred at 110°C overnight. After cooling to room temperature, the mixture was filtered and extracted with EtOAc (3 x 30 mL). The combined organic layers were dried over MgSO4 and concentrated. The crude product was purified by flash column chromatography (SiO2, hexane/EtOAc) to afford the desired product. [4]
Pharmaceutical Applications
Tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate belongs to the class of piperazine compounds, which are important nitrogen-containing heterocycles with unique structures and diverse applications, widely utilized in fields such as materials, dyes, and pharmaceuticals. Studies have reported that tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate can be employed in the synthesis of the drug molecule Palbociclib. Palbociclib, marketed under the brand name Ibrance and developed by Pfizer, is a medication indicated for the treatment of hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative breast cancer. It functions as a selective inhibitor of the cyclin-dependent kinases CDK4 and CDK6, and it holds the distinction of being the first CDK4/6 inhibitor approved for cancer therapy. Palbociclib was reviewed and granted accelerated approval by the U.S. Food and Drug Administration on February 3, 2015, under the Priority Review and Breakthrough Therapy designation programs. It is approved for use in combination with letrozole for the treatment of patients with estrogen receptor-positive (ER+) advanced breast cancer.
Reference
[1] Chen, Weijiao; et al, Discovery, Optimization, and Evaluation of Selective CDK4/6 Inhibitors for the Treatment of Breast Cancer, Journal of Medicinal Chemistry 2022, 65, 15102-15122.
[2] Li S, Chen J, Feng C, et al. A new route for the synthesis of Palbociclib[J]. Chemical Papers, 2019, 73: 3043-3051.
[3] Shi, Wei; et al, Discovery of Potent and Selective CDK4/6 Inhibitors for the Treatment of Chemotherapy-Induced Myelosuppression, Journal of Medicinal Chemistry 2025, 68, 1446-1472.
[4] Li, Yi-Qian; et al, Discovery of Novel Bruton's Tyrosine Kinase PROTACs with Enhanced Selectivity and Cellular Efficacy, Journal of Medicinal Chemistry (2023), 66(11), 7454-7474.
You may like
See also
Lastest Price from tert-butyl 4-(6-aminopyridin-3-yl)piperazine-1-carboxylate manufacturers
US $0.00-0.00/kg2025-08-29
- CAS:
- 571188-59-5
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 1

US $0.00-0.00/Kg/Bag2025-04-21
- CAS:
- 571188-59-5
- Min. Order:
- 2Kg/Bag
- Purity:
- 0.98
- Supply Ability:
- 20 tons