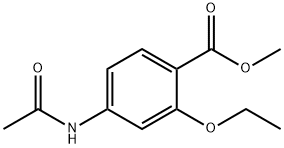
Chemical Structure and Pharmacological Effects of Ethopabate
Ethopabate is a common feed additive primarily used as an anticoccidial agent in veterinary medicine. Ethopabate exerts its effects by blocking the growth and reproduction of coccidia, thereby protecting animals from coccidiosis. Ethopabate contains a benzoate group in its chemical structure, which allows it to interfere with the metabolic processes of coccidia. Ethopabate acts on the tetrahydrofolate synthesis pathway of coccidia by competing with PABA (para-aminobenzoic acid) for the enzyme dihydropteroate synthase, thereby inhibiting the production of dihydrofolate and subsequently affecting the growth and reproduction of coccidia. Additionally, Ethopabate can be used in combination with other anticoccidial drugs such as amprolium to enhance therapeutic efficacy and compensate for the limitations of a single drug.

Figure1: Picture of Ethopabate
Characteristics
As a veterinary drug additive, Ethopabate is mainly used in animal husbandry for the prevention and treatment of coccidiosis in poultry such as chickens and ducks. Ethopabate exhibits strong efficacy against Eimeria maxima, E. brunetti, and other small intestinal coccidia. In particular, Ethopabate effectively compensates for the shortcomings of amprolium against coccidia species where amprolium has weaker effects. Although Ethopabate has insufficient activity against Eimeria tenella, when combined with amprolium, the two agents complement each other to achieve better anticoccidial effects. Ethopabate is characterized by high efficiency, low toxicity, and no mutagenic or carcinogenic effects. Moreover, Ethopabate does not accumulate in poultry, making it relatively safe for both animals and humans. The use of Ethopabate not only prevents and treats coccidiosis but also reduces feed consumption by coccidia, improves feed conversion efficiency, and promotes the growth and development of poultry. [1]
Chemical Structure
From a chemical structure perspective, coccidiostat esters mainly include polyether coccidiostats and ionophore coccidiostats. Ethopabate, as a representative of the polyether type, is represented by monensin ester, which is obtained by the esterification of polyether compounds produced by microbial fermentation. Ethopabate of the monensin ester type contains multiple ether bonds and ester groups in its molecular structure, giving it lipophilic properties that allow it to easily cross the cell membrane of coccidia. Ethopabate can also be exemplified by the ionophore type, such as lasalocid sodium ester, whose molecular structure contains a special ionophore structure capable of forming complexes with metal ions (e.g., sodium and potassium ions), thereby interfering with the ionic balance within coccidia cells. [1]
Efficacy and Characteristics
In terms of efficacy and characteristics, different types of coccidiostat esters exhibit their respective advantages. Ethopabate, represented by the polyether type monensin ester, demonstrates good inhibitory effects against various coccidia species, particularly Eimeria tenella and Eimeria necatrix. Ethopabate of this type achieves its anticoccidial purpose by disrupting the ionic gradient within coccidial cells, thereby preventing normal material exchange and energy metabolism, which inhibits the growth and reproduction of coccidia. Additionally, Ethopabate in the form of monensin ester possesses certain growth-promoting effects, promoting the growth and development of animals while preventing coccidiosis. On the other hand, Ethopabate as the ionophore type lasalocid sodium ester exhibits stronger selectivity against coccidia, primarily acting on the trophozoite stage of coccidia and inhibiting their development by affecting cation transport within coccidial cells. Ethopabate in the form of lasalocid sodium ester achieves significant anticoccidial effects at low doses and is less prone to inducing drug resistance.
Pharmacological Effects
Ethopabate, when used in combination with other drugs, can not only inhibit the shedding of infected Eimeria maxima oocysts in chickens and exert anticoccidial effects by blocking the synthesis of tetrahydrofolate in the PABA-folate metabolic pathway, but also effectively delay the development of drug resistance. Ethopabate is characterized by high efficacy, non-toxicity, non-mutagenicity, non-carcinogenicity, and no accumulation in poultry.
Biological Applications
In the application of anticoccidial drugs, different types of coccidiostat esters, including Ethopabate, are widely used in various formulations. Ethopabate in the form of monensin ester is often made into premixes and added to animal feed for convenient intake, thereby preventing and treating coccidiosis. It is widely used in the farming of poultry and livestock such as chickens, ducks, and rabbits, effectively controlling coccidial infections and improving farming efficiency. Ethopabate as lasalocid sodium ester, in addition to being formulated as a premix, can also be prepared as a soluble powder for drinking water administration, suitable for different farming environments and methods. It is commonly used in large-scale chicken farms to precisely control coccidial infections and ensure the healthy growth of chicken flocks. [2]
Reference
[1] Gao F, Yin C X, Huo F J, et al. Ethopabate[J]. Structure Reports, 2005, 61(11): 3870-3871.
[2] Szalkowski C R. Ethopabate in feeds[J]. Journal of the Association of Official Agricultural Chemists, 1965, 48: 280-284.
You may like
See also
Lastest Price from Ethopabate manufacturers

US $0.00-0.00/Kg/Drum2025-04-21
- CAS:
- 59-06-3
- Min. Order:
- 1KG
- Purity:
- 99%min
- Supply Ability:
- 500kg

US $10.00/kg2025-04-21
- CAS:
- 59-06-3
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 10 mt