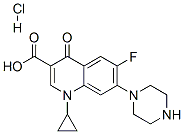
Ciprofloxacin Hydrochloride: Clinical Uses & Toxicity
Ciprofloxacin hydrochloride is used to treat or prevent certain infections caused by bacteria such as pneumonia; gonorrhea (a sexually transmitted disease); typhoid fever (a serious infection that is common in developing countries); infectious diarrhea (infections that cause severe diarrhea); and infections of the skin, bone, joint, abdomen (stomach area), and prostate (male reproductive gland), it is also used to treat or prevent plague (a serious infection that may be spread on purpose as part of a bioterror attack) and inhalation anthrax (a serious infection that may be spread by anthrax germs in the air on purpose as part of a bioterror attack). Ciprofloxacin hydrochloride may also be used to treat bronchitis, sinus infections, or urinary tract infections but should not be used for bronchitis and sinus infections, or certain types of urinary tract infections if there are other treatment options.

Resistance mechanisms of bacteria against ciprofloxacin
A member of the fluoroquinolone (FQ) family of antibiotics, ciprofloxacin hydrochloride can be used to treat a variety of Gram-positive and Gram-negative bacteria. FQs regulate bacterial DNA supercoiling, a procedure necessary for DNA replication, recombination, and repair, by binding to and inhibiting DNA gyrase enzymes. Nonetheless, there are increasing reports of ciprofloxacin resistance in Bacillus anthracis, Pseudomonas aeruginosa, Neisseria gonorrhoeae, Enterococci, Escherichia coli, and Klebsiella pneumoniae. The resistance could develop by efflux pumps or mutations in DNA gyrase genes (gyrA). Ciprofloxacin hydrochloride can also be used in the treatment of malaria. In this regard, the review mainly concentrated on the various properties of ciprofloxacin, its clinical applications for the treatment of different microbial infections, and bacterial resistance mechanisms to this antibiotic, as well as new strategies for enhancing ciprofloxacin efficacy against MDR bacteria. Treatment failures may occur due to microbial resistance to effective broad-spectrum antibiotics. Treatment failures and difficult-to-treat infections could lead to a high death rate. Drug target mutations (DNA gyrase and DNA topoisomerase IV), mutations that limit drug accumulation, and plasmids that shield cells from ciprofloxacin's deadly effects are the three mechanisms of ciprofloxacin hydrochloride resistance that have been found.[1]
Ciprofloxacin hydrochloride's potential for the treatment of a large spectrum of bacterial infections led to the overuse of this drug in clinical practice and developed alarming levels of ciprofloxacin resistance as a consequence of heavy use. To preserve this beneficial agent, prescribers must ensure that ciprofloxacin is a proper choice and administer enough doses to limit the risk of selecting resistant mutant bacterial subpopulations. The increasing incidence of ciprofloxacin-resistant pathogens jeopardizes the continued empiric use of ciprofloxacin and raises the urgent need to develop novel ciprofloxacin hydrochloride derivatives potent against both drug-susceptible and drug-resistant pathogens and discover useful synergism between ciprofloxacin and other antibacterial agents. As mentioned earlier in this study, recent studies have reported a wide range of synergism between ciprofloxacin and other antibacterial agents. Therefore, the combination use of ciprofloxacin and other antibiotics and antibacterial agent should be considered in future studies because combination therapy could increase antibacterial performance of ciprofloxacin hydrochloride especially against MDR strains.
Liver Toxicity of Ciprofloxacin hydrochloride
Like other fluoroquinolones, ciprofloxacin hydrochloride is active against a wide range of aerobic gram-positive and gram-negative organisms. The fluoroquinolones are believed to act by inhibition of bacterial DNA gyrase and topoisomerase IV that are required for synthesis of bacterial mRNAs (transcription) and DNA replication. In contrast, DNA gyrases are not present in human [and other eukarotic] cells and the equivalent topoisomerases are not sensitive to fluoroquinolone inhibition. Ciprofloxacin hydrochloride was approved for use in the United States in 1990 and, currently, more than 4 million prescriptions are filled yearly. Ciprofloxacin hydrochloride like other fluoroquinolones is associated with a low rate (1% to 3%) of serum enzyme elevations during therapy. These abnormalities are generally mild, asymptomatic and transient, resolving even with continuation of therapy. More importantly, ciprofloxacin has been linked to rare, but occasionally severe and even fatal cases of acute liver injury. Severity ranges from mild and transient serum enzyme elevations to a self-limited hepatitis, to prolonged cholestatic hepatitis to a fulminant hepatic failure. If not fatal during the acute phase, complete recovery is expected after stopping the drug and is usually rapid (2 to 4 weeks) depending upon the severity and degree of cholestasis. Some instances of cholestatic liver injury from ciprofloxacin hydrochloride have resulted in vanishing bile duct syndrome.[2]
References
[1]Shariati A, Arshadi M, Khosrojerdi MA, Abedinzadeh M, Ganjalishahi M, Maleki A, Heidary M, Khoshnood S. The resistance mechanisms of bacteria against ciprofloxacin and new approaches for enhancing the efficacy of this antibiotic. Front Public Health. 2022 Dec 21;10:1025633. doi: 10.3389/fpubh.2022.1025633. PMID: 36620240; PMCID: PMC9815622.
[2]LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-. Ciprofloxacin. [Updated 2024 May 9].
You may like
Lastest Price from Ciprofloxacin hydrochloride manufacturers

US $1.00/KG2025-04-21
- CAS:
- 93107-08-5
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt

US $45.00/kg2025-04-21
- CAS:
- 93107-08-5
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 20 tons